Advertisements
Advertisements
प्रश्न
Which of the following species cannot act as a ligand? Give reason.
पर्याय
OH−
`"NH"_4^+`
CH3NH2
H2O
उत्तर
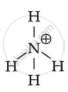
`bb("NH"_4^+)`
Explanation:
Because it is unable of providing a core metal ion with a lone pair of electrons.
APPEARS IN
संबंधित प्रश्न
Write the structure and IUPAC names of all the metamers represented by formula C4H10
What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is passed through this solution?
Write IUPAC names of the following compounds
Write the structures of compounds A, B and C in the following reactions
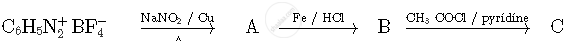
What is the coordination number of chromium in \[\ce{[Cr(NH3)2(H2O2)2]Cl3}\]?
Which of the following is non-ionizable?
The stabilisation of coordination compounds due to chelation is called the chelate effect. Which of the following is the most stable complex species?
Which of the following complexes are homoleptic?
(i) \[\ce{[Co(NH3)6]^{3+}}\]
(ii) \[\ce{[Co(NH3)4 Cl2]^{+}}\]
(iii) \[\ce{[Ni(CN)4]^{2-}}\]
(iv) \[\ce{[Ni(NH3)4Cl2]}\]
The oxidation state of Fe in the brown ring complex [F3(H2O)5NO]SO4 is
Explain the following, giving two examples:
Coordination number
