Advertisements
Advertisements
प्रश्न
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Moles: 1 mole + ______ `→` ______ + ______
उत्तर
1 mole + 2 mole `→` 1 mole + 2 mole
APPEARS IN
संबंधित प्रश्न
What do you understand by exothermic reactions?
Write symbolic representation for the following word equation and balance them :
Carbon + Oxygen → Carbon dioxide.
Write symbolic representation for the following word equation and balance them :
Iron + Sulphur → Iron sulphide.
Complete the following equation:
CH3COOH + NaOH →
Balance the equation stepwise.
SO2(g) + H2S(aq) → S(s) + H2O(l)
Choose the correct answer from the options given below.
The formula of sodium carbonate is Na2CO3 and that of calcium hydrogen carbonate is
Balance the following simple equation:
N2 + O2 ⇌ NO
Fill in the blank with the correct word from the options given below:
A chemical equation is a shorthand form for a _______ change.
Give a balanced equation by the partial equation method, [steps are given below].
Oxidation of Lead [II] Sulphide by Ozone
- Ozone first decomposes to give molecular oxygen & nascent oxygen.
- Nascent oxygen then oxidises lead [II] sulphide to lead [II] sulphate.
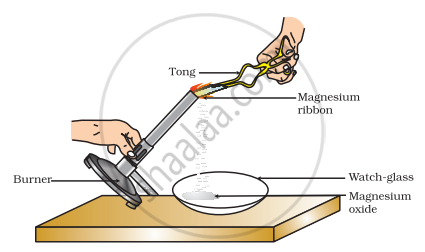
Which of the following is the correct observation of the reaction shown in the above set up?
