Advertisements
Advertisements
प्रश्न
Define the following.
Boiling point
उत्तर
The temperature at which water boils and changes to steam is called as boiling point. The boiling point of water is 100°C at atmospheric pressure.
APPEARS IN
संबंधित प्रश्न
State whether the following statement is true or false.
The substance in which a solute dissolves is called a solvent.
Explain the picture in your own words.
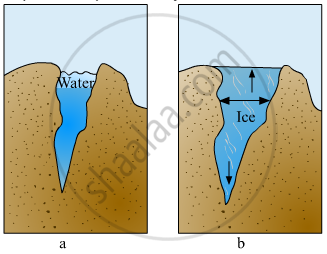
If same mass of liquid water and a piece of ice is taken, then why is the density of ice less than that of liquid water?
\[\ce{H2O2}\] is a better oxidising agent than water. Explain.
Explain why \[\ce{HCl}\] is a gas and \[\ce{HF}\] is a liquid.
Why does water show high boiling point as compared to hydrogen sulphide? Give reasons for your answer.
Write two reactions to explain amphoteric nature of water.
Pure water boils at ______ °C at one atmospheric pressure.
______ has the highest latent heat of fusion.
The density of gold is 19 g/cm3. If 1.9 × 10−4 g of gold is dispersed in one litre of water to give a sol having spherical gold particles of radius 10 nm, the number of gold particles per mm3 of the sol will be ______ × 106.
