Advertisements
Advertisements
प्रश्न
Derive the relationship between density of substance, its molar mass, and the unit cell edge length. Explain how you will calculate the number of particles, and a number of unit cells in x g of metal.
उत्तर
- Relationship between density of a substance, its molar mass and the unit cell edge length:
- If edge length of cubic unit cell is ‘a’, then the volume of unit cell is a3.
- Suppose that mass of one particle is ‘m’ and that there are ‘n’ particles per unit cell.
∴ Mass of unit cell = m × n .....(1) - The density of unit cell (ρ), which is same as density of the substance is given by:
`rho = "Mass of unit cell"/"Volume of a unit cell"`
= `("m" xx "n")/"a"^3` = Density of substance …(2) - Molar mass (M) of the substance is given by:
M = mass of one particle × number of particles per mole
= m × NA (NA is Avogadro number)
Therefore, m = `"M"/"N"_"A"` .....(3) - Combining equations (2) and (3), gives
`rho = ("n M")/("a"^3 "N"_"A")` .....(4)
- The density (ρ) and molar mass (M) of metal are related to each other through unit cell parameters as given below:
`rho = "n"/"a"^3 xx "M"/"N"_"A"`
∴ M = ρ`("a"^3 "N"_"A")/"n"`
where, ‘n’ is the number of particles in unit cell and ‘a3’ is the volume of unit cell.
- The number of particles in x g of metallic crystal:
∴ Molar mass, M, contains NA particles.
∴ x g of metal contains `(x"N"_"A")/"M"` particles.
Substitution of M in the above equation gives
Number of particles in ‘x’ g = `(x"N"_"A")/(rho"a"^3"N"_"A"//"n") = "x n"/(rho "a"^3)` - Number of unit cells in x g of metallic crystal:
‘n’ particles correspond to 1 unit cell.
∴ `(x"n")/(rho"a"^3)` particles correspond to `(x"n")/(rho"a"^3) xx 1/"n"` unit cells.
∴ Number of unit cells in ‘x’ g metal = `x/(rho"a"^3)`
संबंधित प्रश्न
Answer the following in brief.
Calculate the number of atoms in fcc unit cell.
An element with molar mass 27 g/mol forms a cubic unit cell with edge length of 405 pm. If the density of the element is 2.7 g/cm3, what is the nature of the cubic unit cell?
Give the percentage of empty space in bcc lattice.
The pyranose structure of glucose is ____________.
Silver crystallises in fcc structure, if edge length of unit cell is 316.5 pm. What is the radius of silver atom?
An element (atomic mass M g/mol) having bcc structure has unit cell edge 400 pm. The density of the element is ____________ g/cm3.
[NA = 6.0 × 1023 atom mol−1)
How many total constituent particles are present in simple cubic unit cell?
A metal crystallises in bcc unit cell with edge length 'a'. What will be the volume of one atom?
If the edge of a body-centred unit cell is 360 pm, what will be the approximate radius of the atom present in it? (in pm)
Sodium crystallizes in bcc structure with radius 1.86 × 10−8 cm. What is the length of unit cell of sodium?
An element crystallizes bcc type of unit cell, the density and edge length of unit cell is 4 g cm−3 and 500 pm respectively. What is the atomic mass of an element?
An element with density 2.8 g cm−3 forms fcc unit cell having edge length 4 × 10−8 cm. Calculate molar mass of the element.
Copper and silver have ____________ crystal structure.
Consider the following unit cell.
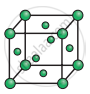
The number of particles (spheres) per unit cell is:
A metallic element has a cubic lattice with edge length of unit cell 2 Å. Calculate the number of unit cells in 200 g of the metal, if density of metal is 2.5 g cm-3?
What is the density of iron crystal which crystallizes in body-centred cubic structure with edge length 287 pm? (At. mass of Fe = 56 amu)
An element has a bee structure with unit cell edge length of 288 pm. How many unit cells and number of atoms are present in 200 g of the element?
Calculate the density of metal with molar mass 56 g mol- 1 that crystallises to form a bcc structure with edge length 288 pm.
Identify unit cell from following having four particles in it
An element with molar mass 2.7 × 10-2 kg/mol. Forms a cubic units cell with edge length of 405 pm. If the density is 2.7 × 103 kg/m3. Find the nature of a cubic unit cell.
At room temperature, polonium Crystallises in a primitive cubic unit cell. If a = 3.36 Å. Calculate the theoretical density of polonium. [It's atomic weight is 209 g/mol.]
Silver crystallizes in the fcc structure. If the edge length of the unit cell is 400 pm, calculate the density of silver (Atomic mass of Ag = 108).
The correct sequence of the atomic layers in cubic close packing is ______.
The total number of different primitive unit cells is ______.
An element crystallises in fee structure. If molar mass of element is 72.7 g mol -1, the mass of its one unit cell will be ______.
