Advertisements
Advertisements
प्रश्न
Describe the role played in the extraction of aluminum:
Sodium hydroxide
उत्तर
In the extraction of aluminium, the given compounds play the following roles:
Sodium hydroxide :
Two roles are played by sodium hydroxide in the extraction of aluminium.
First, finely grinded bauxite (ore of aluminium) is heated under pressure with conc. caustic soda solution (NaOH solution) for 2-8 hours at 140°C to 150°C to produce sodium aluminate. The chemical equation is as follows:
Al2O3.2H2O + 2NaOH → 2NaAlO2+ 3H2O
Second, on diluting sodium aluminate with water and cooling to 50°C, sodium aluminate is hydrolysed to give aluminium hydroxide as a precipitate. Here, the impurities dissolve in sodium hydroxide.
APPEARS IN
संबंधित प्रश्न
Answer the following questions with respect to the electrolytic process in the extraction of aluminum:
Identify the components of the electrolyte other than pure alumina and the role played by each
Name the following: A compound added to lower the fusion temperature of electrolytic bath in the extraction of aluminum
Name the constituents of Solder.
In order to obtain 1 tonne of aluminium, the following inputs are required: 4 tonnes of bauxite, 150 kg of sodium hydroxide and 600 kg of graphite. The aluminium compound in bauxite is aluminium oxide and the main impurity is iron (III) oxide. Aluminium is obtained by the electrolysis of aluminium oxide dissolved in cryolite.
When bauxite is treated with sodium hydroxide solution, what happens to:
- the aluminium oxide,
- the iron (III) oxide?
Name the following:
The substance added along with aluminium in the Hall-Heroult's process.
Write the equation for the reaction where the aluminium oxide for the electrolytic extraction of aluminium is obtained by heating aluminium hydroxide.
Explain why it is preferably to use a number of graphite electrodes as anode instead of a single electrode, during the above electrolysis.
In Hoope's process, pure aluminium is collected at the ______ of the electrolytic cell.
Name the alloy used for the following purpose.
Making parts of watches
The following sketch illustrates the process of conversion of Alumina to Aluminium:
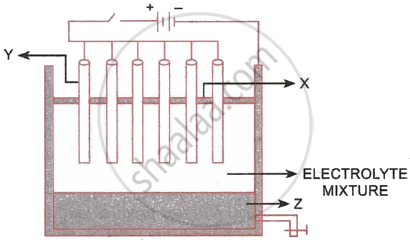
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
