Advertisements
Advertisements
प्रश्न
Describe the roles of the following: SiO2 in the extraction of copper from copper matte
उत्तर
In the extraction of copper from copper matte, SiO2 is used to remove iron oxide as slag
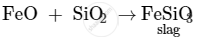
APPEARS IN
संबंधित प्रश्न
What is the chemical composition of malachite?
- CuO . CuC03
- Cu(OH)2 CuC03
- CuO.Cu(OH)2
- Cu2O . Cu(OH)2
Out of C and CO, which is a better reducing agent at the lower temperature range in the blast furnace to extract iron from the oxide ore?
What is the role of cryolite in the extraction of aluminium?
Name the method used for refining of copper metal.
Which among the following minerals does NOT contain aluminium?
(a) Cryolite
(b) Siderite
(c) China clay
(d) Corundum
What is the role of the following compound-
SiO2 in the extraction of copper from copper pyrites?
In the extraction of aluminium by Hall-Heroult process, purified \[\ce{Al2O3}\] is mixed with \[\ce{CaF2}\] to:
(i) lower the melting point of \[\ce{Al2O3}\].
(ii) increase the conductivity of molten mixture.
(iii) reduce \[\ce{Al^{3+}}\] into \[\ce{Al(s)}\].
(iv) acts as catalyst.
When a strip of metallic zinc is placed in an aqueous solution of copper nitrate the blue colour of the solution disappear due to formation of
The important ore of iron is:-
When compared to ΔG° for the formation of Al2O3 ΔG° for the formation of Cr2O3 is
