Advertisements
Advertisements
Question
Describe the roles of the following: SiO2 in the extraction of copper from copper matte
Solution
In the extraction of copper from copper matte, SiO2 is used to remove iron oxide as slag
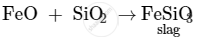
APPEARS IN
RELATED QUESTIONS
Draw neat and labelled diagram of Bessermer converter used in the extraction of copper.
What is the role of CO in the extraction of Iron?
Which form of the iron is the purest form of commercial iron?
Draw a neat, well labelled diagram of electrolytic cell for extraction of aluminium
What are the differences between minerals and ores?
The most abundant metal on earth is:
The main reactions occurring in blast furnace during extraction of iron from haematite are:
(i) \[\ce{Fe2O3 + 3CO -> 2Fe + 3CO2}\]
(ii) \[\ce{FeO + SiO2 -> FeSiO3}\]
(iii) \[\ce{Fe2O3 + 3C -> 2Fe + 3CO}\]
(iv) \[\ce{CaO + SiO2 -> CaSiO3}\]
When compared to ΔG° for the formation of Al2O3 ΔG° for the formation of Cr2O3 is
Which of the following statements, about the advantage of roasting of sulphide ore before reduction is not true?
Match List I with List II.
| List - I | List - II | ||
| a. | Siderite | I. | FeCO3 |
| b. | Malachite | II. | CuCO3 · Cu(OH)2 |
| c. | Sphalerite | III. | ZnS |
| d. | Calamine | IV. | ZnCO3 |
Choose the correct answer from the option given below.
