Advertisements
Advertisements
Questions
Write the names and structure of the monomers of the following polymers: Nylon-6, 6
Write the names of monomers used for getting the following polymers:
Nylon-6,6
Solution 1
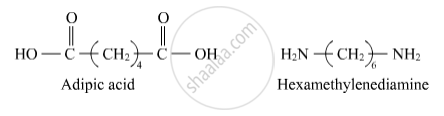
Solution 2
The monomers of nylon-6,6 are hexamethylenediamine and adipic acid.
RELATED QUESTIONS
Write the names of monomers of the following polymers:
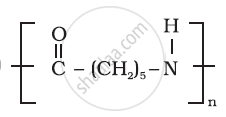
How can you differentiate between addition and condensation polymerisation?
How is dacron obtained from ethylene glycol and terephthalic acid?
Write a note on aldol condensation.
Name the monomers and the type of polymerization in each of the following polymers:
Bakelite
What is the significance of number of 6,6 in the polymer nylon-6,6?
Which of the following polymers can have strong intermolecular forces?
(i) Nylon
(ii) Polystyrene
(iii) Rubber
(iv) Polyesters
Assertion: Polyamides are best used as fibres because of high tensile strength.
Reason: Strong intermolecular forces (like hydrogen bonding within polyamides) lead to close packing of chains and increase the crystalline character, hence, provide high tensile strength to polymers
Acetic acid is added in the preparation of nylon-6 to
The polymer containing strong intermolecular forces, e.g., hydrogen bonding, is ______.
