Advertisements
Advertisements
Question
Which of the following polymers can have strong intermolecular forces?
(i) Nylon
(ii) Polystyrene
(iii) Rubber
(iv) Polyesters
Solution
(i) Nylon
(iv) Polyesters
Explanation:
Nylon and polyester are thread forming fibres which possesses high tensile strength and high melting point. They have strong intermolecular forces like hydrogen bonding.
APPEARS IN
RELATED QUESTIONS
Write the names and structure of the monomers of the following polymers: Nylon-6, 6
Which of the following is a fibre? Nylon, neoprene, PVC
Write the names of monomers of the following polymers.
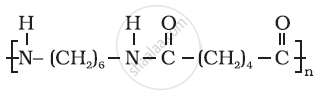
Name the monomers and the type of polymerization in each of the following polymers:
Bakelite
What is the significance of number of 6,6 in the polymer nylon-6,6?
Write the name of the monomer of the following polymer:
Bakelite
Write the structures of monomers used the following polymers:
Nylon - 6, 6
Assertion: Polyamides are best used as fibres because of high tensile strength.
Reason: Strong intermolecular forces (like hydrogen bonding within polyamides) lead to close packing of chains and increase the crystalline character, hence, provide high tensile strength to polymers
The polymer formed on heating Novolac with formaldehyde is ______.
Match List - I with List - II:
| List - I (Polymer) |
List - II (used for items) |
||
| (A) | Nylon 6, 6 | (I) | Buckets |
| (B) | Low density polythene | (II) | Non-stick utensils |
| (C) | High density polythene | (III) | Bristles of brushes |
| (D) | Teflon | (IV) | Toys |
Choose the correct answer from the options given below:
