Advertisements
Advertisements
प्रश्न
Which of the following polymers can have strong intermolecular forces?
(i) Nylon
(ii) Polystyrene
(iii) Rubber
(iv) Polyesters
उत्तर
(i) Nylon
(iv) Polyesters
Explanation:
Nylon and polyester are thread forming fibres which possesses high tensile strength and high melting point. They have strong intermolecular forces like hydrogen bonding.
APPEARS IN
संबंधित प्रश्न
Write the names of monomers of the following polymers:
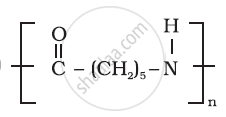
How can you differentiate between addition and condensation polymerisation?
Name the monomers and the type of polymerization in each of the following polymers:
Bakelite
What is the significance of number of 6,6 in the polymer nylon-6,6?
Which factor imparts crystalline nature to a polymer like nylon?
Match the polymers given in Column I with their main applications given in Column II.
| Column I | Column II |
| (i) Bakelite | (a) Unbreakable crockery |
| (ii) Low-density polythene | (b) Non-stick cookwares |
| (iii) Melamine-formaldehyderesin | (c) Packaging material for shock absorbance |
| (iv) Nylon 6 | (d) Electrical switches |
| (v) Polytetrafluoroethane | (e) Squeeze bottles |
| (vi) Polystyrene | (f) Tyre, cords |
Assertion: Polyamides are best used as fibres because of high tensile strength.
Reason: Strong intermolecular forces (like hydrogen bonding within polyamides) lead to close packing of chains and increase the crystalline character, hence, provide high tensile strength to polymers
Which of the following polymer is used for making phonograph records?
A raw material used in making nylon is
Which is used for the formation of nylon – 6?
