Advertisements
Advertisements
Question
Draw neat and labelled diagram of Bessermer converter used in the extraction of copper.
Solution 1
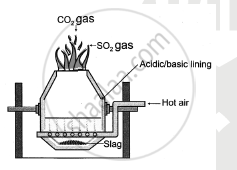
Solution 2

APPEARS IN
RELATED QUESTIONS
Write the names and chemical formulae of any one ore of iron.
What is the chemical composition of malachite?
- CuO . CuC03
- Cu(OH)2 CuC03
- CuO.Cu(OH)2
- Cu2O . Cu(OH)2
Write the reactions involved in the zone of reduction in blast furnace during extraction of iron.
Write chemical formulae of the following ores :
(a) Calamine
(b) Haematite
(c) Magnetite
(d) Corundum
Out of C and CO, which is a better reducing agent at the lower temperature range in the blast furnace to extract iron from the oxide ore?
What is the role of limestone in the extraction of iron from its oxides?
Name the method used for refining of copper metal.
Draw a neat, well labelled diagram of electrolytic cell for extraction of aluminium
Why does copper obtained in the extraction from copper pyrites have a blistered appearance?
What is the role of the following compound-
SiO2 in the extraction of copper from copper pyrites?
What happens when a nickel rod is dipped into a copper sulphate solution? Justify your answer.
`[E_((Ni^(+2))/(Ni))^0 = -0.25"v" and E _(("Cu"^(+2))/("Cu"))^0 = +0.34"v"]`
Describe the role of the Cryolite in the extraction of aluminium from pure alumina.
Which among the following statement is correct?
The sulphide ore of iron is ____________.
Carbonate ore of iron is ____________.
When copper ore is mixed with silica, in a reverberatory furnace copper matte is produced. The copper matte contains ______.
In the extraction of aluminium by Hall-Heroult process, purified \[\ce{Al2O3}\] is mixed with \[\ce{CaF2}\] to:
(i) lower the melting point of \[\ce{Al2O3}\].
(ii) increase the conductivity of molten mixture.
(iii) reduce \[\ce{Al^{3+}}\] into \[\ce{Al(s)}\].
(iv) acts as catalyst.
Which of the following reactions occur during calcination?
(i) \[\ce{CaCo3 -> CaO + CO2}\]
(ii) \[\ce{2FeS2 + 11/2 O2 -> Fe2O3 + 4SO2}\]
(iii) \[\ce{Al2O3. {x}H2O -> Al2O3 + {x}H2O}\]
(iv) \[\ce{ZnS + 3/2 O2 -> ZnO + SO2}\]
The main reactions occurring in blast furnace during extraction of iron from haematite are:
(i) \[\ce{Fe2O3 + 3CO -> 2Fe + 3CO2}\]
(ii) \[\ce{FeO + SiO2 -> FeSiO3}\]
(iii) \[\ce{Fe2O3 + 3C -> 2Fe + 3CO}\]
(iv) \[\ce{CaO + SiO2 -> CaSiO3}\]
When a strip of metallic zinc is placed in an aqueous solution of copper nitrate the blue colour of the solution disappear due to formation of
Metal always found in Free State is
Which of the following is not an ore of magnesiuim?
Match List I with List II.
| List - I | List - II | ||
| a. | Siderite | I. | FeCO3 |
| b. | Malachite | II. | CuCO3 · Cu(OH)2 |
| c. | Sphalerite | III. | ZnS |
| d. | Calamine | IV. | ZnCO3 |
Choose the correct answer from the option given below.
Match List - I with List - II.
| List - I | List - II | ||
| (One) | (Composition) | ||
| (A) | Siderite | (I) | FeCO3 |
| (B) | Malachite | (II) | CuCO3 · Cu(OH)2 |
| (C) | Sphalerite | (III) | ZnS |
| (D) | Calamine | (IV) | ZnCO3 |
Choose the correct answer from the options given below:
Calamine and Malachite, respectively, are the ores of ______.
The ore that contains both iron and copper is ______.
