Advertisements
Advertisements
प्रश्न
Determine the shortest wavelengths of Balmer and Pasch en series. Given the limit for the Lyman series is 912 Å.
उत्तर
The wavelength of lines are given by,
`1/lambda = "R"_"H" (1/"n"^2 - 1/"m"^2)`
For lyman series limit. n = 1. m = ∞
`therefore 1/lambda_"L" = "R"_"H" (1/1^2 - 1/oo) = "R"_"H"`
`therefore lambda_"L" = 1/"R"_"H" = 912`Å
For Balmer series limit, n = 2, m = ∞
`therefore 1/lambda_"B" = "R"_"H" (1/2^2 - 1/oo) = "R"_"H"/4`
`therefore lambda_"B" = 4/"R"_"H"`
= 4 × 912
= 3648 Å
For Paschen series limit. n = 3, m = ∞
`1/lambda_"P" = "R"_"H" (1/3^2 - 1/oo) = "R"_"H"/9`
`therefore lambda_"P" = 9/"R"_"H" = 9 xx 912`
= 8208 Å
APPEARS IN
संबंधित प्रश्न
Determine the series limit of Balmer, Paschen, and Pfund series, given the limit for Lyman series is 912 Å.
In Balmer series, wavelength of first line is 'λ1' and in Brackett series wavelength of first line is 'λ2' then `lambda_1/lambda_2` is ______.
In the figure below, D and E respectively represent
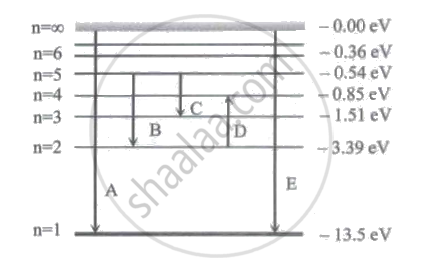
Which of the following is TRUE?
Let the series limit for Balmer series be 'λ1' and the longest wavelength for Brackett series be 'λ2'. Then λ1 and λ2 are related as ______.
Which of the following transition will have highest emission frequency?
The energy (in eV) required to excite an electron from n = 2 to n = 4 state in hydrogen atom is ____________.
In hydrogen atom, the product of the angular momentum and the linear momentum of the electron is proportional to (n = principal quantum number) ____________.
In hydrogen spectrum, which of the following spectral series lies in ultraviolet region?
The radii of the first four Bohr orbits of hydrogen atom are related as ____________.
In hydrogen spectrum, the series of lines obtained in the ultraviolet region of the spectrum is ____________.
If the mass of the electron is reduced to half, the Rydberg constant ______.
Each element is associated with a ______.
Continuous spectrum is produced by ______.
An electron makes a transition from orbit n = 4 to the orbit n = 2 of a hydrogen atom. What is the wave number of the emitted radiations? (R = Rydberg's constant)
To produce an emission spectrum of hydrogen it needs to be ______.
What is the minimum energy that must be given to a H atom in ground state so that it can emit an Hγ line in Balmer series. If the angular momentum of the system is conserved, what would be the angular momentum of such Hγ photon?
The first four spectral lines in the Lyman series of a H-atom are λ = 1218 Å, 1028Å, 974.3 Å and 951.4 Å. If instead of Hydrogen, we consider Deuterium, calculate the shift in the wavelength of these lines.
Deutrium was discovered in 1932 by Harold Urey by measuring the small change in wavelength for a particular transition in 1H and 2H. This is because, the wavelength of transition depend to a certain extent on the nuclear mass. If nuclear motion is taken into account then the electrons and nucleus revolve around their common centre of mass. Such a system is equivalent to a single particle with a reduced mass µ, revolving around the nucleus at a distance equal to the electron-nucleus separation. Here µ = meM/(me + M) where M is the nuclear mass and m e is the electronic mass. Estimate the percentage difference in wavelength for the 1st line of the Lyman series in 1H and 2H. (Mass of 1H nucleus is 1.6725 × 10–27 kg, Mass of 2H nucleus is 3.3374 × 10–27 kg, Mass of electron = 9.109 × 10–31 kg.)
In the hydrogen atoms, the transition from the state n = 6 to n = 1 results in ultraviolet radiation. Infrared radiation will be obtained in the transition.
The de-Broglie wavelength of the electron in the hydrogen atom is proportional to ______.
Calculate the wavelength of the first two lines in the Balmer series of hydrogen atoms.
