Advertisements
Advertisements
प्रश्न
Draw all the isomers (geometrical and optical) of [Co(NH3)2Cl2(en)]+.
उत्तर
(i)
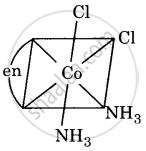
(ii)
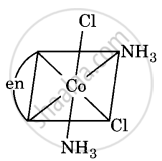
(iii)
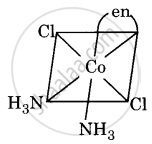
(iv)
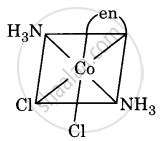
All are asymmetric. Hence, all will show optical isomerism, i.e. d(+) and l(-) forms, which are non-superimposable mirror images.
APPEARS IN
संबंधित प्रश्न
Draw one of the geometrical isomers of the complex [Pt (en)2Cl2] +2 which is optically inactive
Draw the geometrical isomers of complex \[\ce{[Co(en)2Cl2]+}\].
Draw the geometrical isomers of complex [Pt(NH3)2Cl2].
Indicate the types of isomerism exhibited by the following complex and draw the structures for these isomers:
K[Cr(H2O)2(C2O4)2]
How many geometrical isomers are possible in the following coordination entity?
[Co(NH3)3Cl3]
Draw the structure of optical isomers of [PtCl2(en)2]2+.
Name the type of isomerism shown by the following pair of compounds:
[CoCl(H2O)(NH3)4]Cl2 and [CoCl2(NH3)4]Cl.H2O
Write the IUPAC name of [Co(en)2Cl2]+ ion and draw the structures of its geometrical isomers.
The IUPAC name for [CoCl(NO2)(en)2]Cl is ____________.
Name the type of isomerism shown by the following pair of compounds:
[Cr(NH3)5Br]SO4 and [Cr(NH3)5SO4]Br
Which one of the following complexes shows optical isomerism? (en = ethylenediamine)
Which of the following molecules has a chiral centre correctly labelled with an asterisk (*)?
Assertion (A): Trans [CrCl2(ox)2]3− shows optical isomerism.
Reason (R): Optical isomerism is common in octahedral complexes involving didentate ligands.
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers: [Pt(NH3)(H2O)Cl2]
Assertion: Addition of bromine water to 1-butene gives two optical isomers.
Reason: The product formed contains two asymmetric carbon atoms.
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the type of isomerism exhibited by the following complex and draw the structure for the isomer:
\[\ce{[Pt(NH3 )(H2O)Cl2]}\]
