Advertisements
Advertisements
प्रश्न
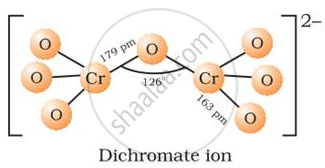
Draw structure of dichromate ion
उत्तर
Dichromate salts contain the dichromate anion, Cr2O72−
They are oxoanions of chromium in the oxidation state +6.
They are moderately strong oxidizing agents.
In an aqueous solution, dichromate ions can be interconvertible.
structure of dichromate ion Cr2O72−:
APPEARS IN
संबंधित प्रश्न
What is the geometry of chromate ion?
- Tetrahedral
- Octahedral
- Trigonal planer
- Linear
What is the molecular formula of chromyl chloride?
(A) CrO2Cl2
(B) CrOCl2
(C) CrCl3
(D) Cr2OCl2
Draw structure of chromate ion
Write observed electronic configuration of elements from first transition series having half
filled d-orbitals.
In acid medium, potassium permanganate oxidizes oxalic acid to ____________.
Permanganate ion changes to ____________ in acidic medium.
How many moles of I2 are liberated when 1 mole of potassium dichromate react with potassium iodide?
The number of moles of acidified KMnO4 required to oxidize 1 mole of ferrous oxalate (FeC2O4) is ____________.
Describe the preparation of potassium dichromate.
Complete the following.
\[\ce{C6H5CH3 ->[acidified][KMnO4]?}\]
Complete the following.
\[\ce{MnO^-_4 + Fe^2+ ->?}\]
Complete the following.
\[\ce{KMnO4 ->[\Delta][Red hot]?}\]
Complete the following.
\[\ce{Cr2O^2-_7 + I^- + H^+ ->?}\]
Complete the following.
\[\ce{Na2Cr2O7 + KCl ->?}\]
Why do the d-block elements form coloured compounds?
