Advertisements
Advertisements
प्रश्न
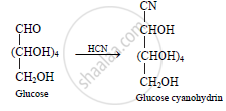
What happens when glucose is treated with hydrogen cyanide?
उत्तर
Action of hydrogen cyanide (HCN) on glucose: The reaction of glucose with
hydrogen cyanide gives cyanohydrin. This indicates the presence of carbonyl group.
APPEARS IN
संबंधित प्रश्न
Write the reaction that indicates the presence of -CHO group in glucose
Write the product when D-glucose reacts with conc. HNO3.
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
What do you observe when glucose solution is heated with Tollen’s reagent?
What do you observe when glucose is treated with bromine water?
Answer the following question.
What is the basic structural difference between glucose and fructose?
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
The spatial arrangement of the given molecule is denoted by:

Choose the appropriate answer(s) for the below representation from the options given
The number of asymmetric carbon atom(s) below the figure is/are

What is the most abundant organic compound on earth?
Acetylation of glucose yields ____________.
Which of the following statements is incorrect regarding glucose?
The symbols D and L represents ____________.
Glucose is found to exist in two different α and β crystalline forms. These forms can be obtained by:
(i) The α form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(ii) The β form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(iii) The β form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
(iv) The α form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
Reduction of glucose by HI suggest that ____________.
The reaction of glucose with red P + HI is called ____________.
Which of the following reactions of glucose can be explained only by its cyclic structure?
The number of chiral carbons in ß-D(+) glucose is ____________.
Which one of the following reactions is not explained by the open chain Structure of glucose?
Which of the following pairs represents anomers?
What happens when D-glucose is treated with the following reagent?
HI
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
Consider the following reactions:
(i) \[\ce{Glucose + R-OH ->[Conc. HNO3] [A] ->[X eq of][(CH3CO)2O] Acetyl derivative}\]
(ii) \[\ce{Glucose ->[Ni/H2] [A] ->[Y eq of][(CH3CO)2O] Acetyl derivative}\]
(iii) \[\ce{Glucose ->[Z eq of][(CH3CO)2O] Acetyl derivative}\]
'X, 'Y' and 'Z' in these reactions are respectively:
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
When D-glucose reacts with HI, it forms ______.
Give a reason for the following observations:
Penta-acetate of glucose does not react with hydroxylamine.
