Advertisements
Advertisements
प्रश्न
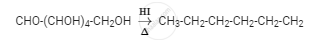
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
उत्तर
The reaction of glucose with HI giving n-hexane suggests that all the six carbon atoms are linked in a straight chain, as shown in the reaction given below:
APPEARS IN
संबंधित प्रश्न
What do you observe when glucose solution is heated with Tollen’s reagent?
What do you observe when glucose is treated with bromine water?
Write the reactions involved when D-glucose is treated with the following reagent:
Br2 water
Write the reactions involved when D-glucose is treated with the following reagent:
(CH3CO)2O
The two forms of D-glucopyranose obtained from the solution of D-glucose are called ____________.
Which one is correct?
In the following reaction, identify A and B:
\[\begin{array}{cc}
\ce{C6H12O6 ->[Acetic anhydride] A}\\
\downarrow \text{Conc. nitric acid}\phantom{...}\\
\ce{B}\phantom{.................}\end{array}\]
Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?
| (I) |  |
| (II) | 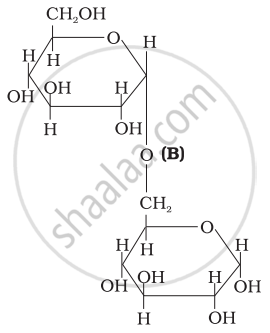 |
| (III) | 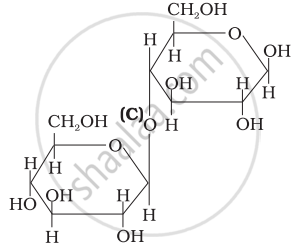 |
Write the reactions of D-glucose which can’t be explained by its open-chain structure. How can cyclic structure of glucose explain these reactions?
Give a reason for the following observations:
Penta-acetate of glucose does not react with hydroxylamine.
