Advertisements
Advertisements
Question
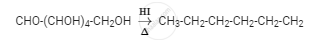
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
Solution
The reaction of glucose with HI giving n-hexane suggests that all the six carbon atoms are linked in a straight chain, as shown in the reaction given below:
APPEARS IN
RELATED QUESTIONS
Enlist the properties of glucose that can not be explained on the basis of open chain structure of it
What happens when glucose is treated with hydrogen cyanide?
Which of the following statements is incorrect regarding glucose?
Glucose reacts with acetic anhydride to form ______.
Glucose is found to exist in two different α and β crystalline forms. These forms can be obtained by:
(i) The α form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(ii) The β form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(iii) The β form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
(iv) The α form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
Reduction of glucose by HI suggest that ____________.
Which is the least stable form of glucose?
The number of chiral carbon atoms present in cyclic structure α-D(+) glucose:
Which of the following pairs represents anomers?
Account for the following:
There are 5 OH groups in glucose
