Advertisements
Advertisements
प्रश्न
Write the reactions of D-glucose which can’t be explained by its open-chain structure. How can cyclic structure of glucose explain these reactions?
उत्तर
Following reactions and facts could not be explained by open-chain structures of glucose.
• Despite having the aldehyde group, glucose does not give 2, 4 − DNP test, Schiff's test and it does not form the hydrogen sulphide addition product with \[\ce{NaHSO3}\].
• The pentaacetate of glucose does not react with hydroxylamine indicating the absence of a free −CHO group.
It was proposed that one of the −OH groups may add to the −CHO group and form a cyclic hemiacetal structure. It was found that glucose forms a six-membered ring in which −OH at C − 5 is involved in a ring formation.
APPEARS IN
संबंधित प्रश्न
How many moles of acetic anhydride will be required to form glucose pentaacetate from 2M of glucose?
(a) 2
(b) 5
(c) 10
(d) 2.5
Write the product when D-glucose reacts with conc. HNO3.
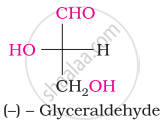
The number of asymmetric carbon atom(s) below the figure is/are

Glucose is found to exist in two different α and β crystalline forms. These forms can be obtained by:
(i) The α form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(ii) The β form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(iii) The β form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
(iv) The α form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
Choose the correct relationship for glucose and fructose:
The letter D and L in carbohydrates represent ____________.
The number of chiral carbons in ß-D(+) glucose is ____________.
Which of the following pairs represents anomers?
Why does compound (A) given below not form an oxime?
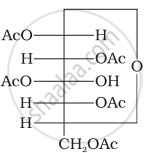
(A)
