Advertisements
Advertisements
प्रश्न
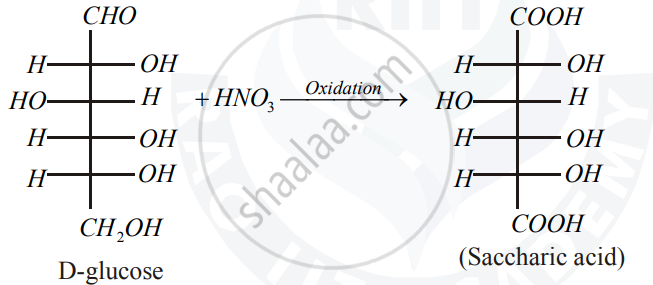
Write the product when D-glucose reacts with conc. HNO3.
उत्तर १
When D-Glucose is treated with nitric acid, the aldehyde and alcohol group of glucose are oxidized to carboxylic acids forming D-Glucaric acid
उत्तर २
`"D-glucose" + HNO_3 ->??`
APPEARS IN
संबंधित प्रश्न
Write the reaction that indicates the presence of -CHO group in glucose
Glucose on reaction with HI gives n-hexane. What does it suggest about the structure of glucose?
The spatial arrangement of the given molecule is denoted by:

When glucose reacts with bromine water, the main product is ____________.
Reduction of glucose by HI suggest that ____________.
Why does compound (A) given below not form an oxime?
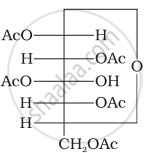
(A)
What happens when D-glucose is treated with the following reagent?
HI
Account for the following:
There are 5 OH groups in glucose
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
