Advertisements
Advertisements
प्रश्न
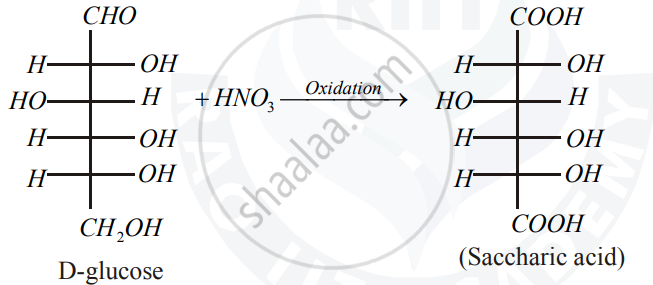
Write the product when D-glucose reacts with conc. HNO3.
उत्तर १
When D-Glucose is treated with nitric acid, the aldehyde and alcohol group of glucose are oxidized to carboxylic acids forming D-Glucaric acid
उत्तर २
`"D-glucose" + HNO_3 ->??`
APPEARS IN
संबंधित प्रश्न
Differentiable between the following:
Amylose and Amylopectin
What do you observe when glucose is treated with bromine water?
Acetylation of glucose yields ____________.
The reaction of glucose with red P + HI is called ____________.
Which is the least stable form of glucose?
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
The two forms of D-glucopyranose obtained from the solution of D-glucose are called ____________.
Which one of the following reactions is not explained by the open chain Structure of glucose?
What happens when D-glucose is treated with the following reagent?
HI
Match List - I with List - II.
| List I | List II | ||
| (A) | Glucose + HI | (I) | Gluconic acid |
| (B) | Glucose + Br2 water | (II) | Glucose pentacetate |
| (C) | Glucose + acetic anhydride | (III) | Saccharic acid |
| (D) | Glucose + HNO3 | (IV) | Hexane |
Choose the correct answer from the options given below:
