Advertisements
Advertisements
प्रश्न
Why does compound (A) given below not form an oxime?
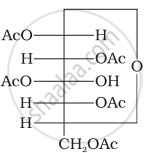
(A)
उत्तर
Glucose pentaacetate (structure A) does not have a free –OH group at C, and therefore, cannot be converted to the open chain form to give a free –CHO group and hence it does not form the oxime.
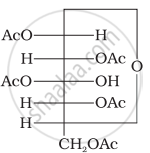
(Structure A)
APPEARS IN
संबंधित प्रश्न
Write the reaction that indicates the presence of -CHO group in glucose
What happens when glucose is treated with hydroxylamine?
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
The following compound can be called as:
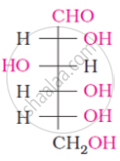
The spatial arrangement of the given molecule is denoted by:

The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?
| (I) |  |
| (II) | 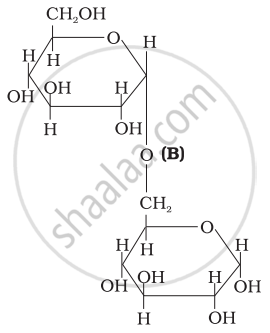 |
| (III) | 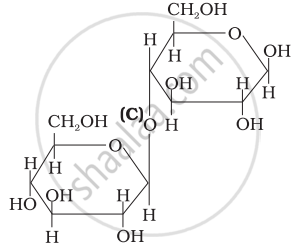 |
Assertion: D (+) – Glucose is dextrorotatory in nature.
Reason: ‘D’ represents its dextrorotatory nature.
Account for the following:
What happens when D – glucose is treated with the following reagents
HNO3
Glucose with excess of phenyl hydrazine forms ______.
