Advertisements
Advertisements
प्रश्न
Assertion: D (+) – Glucose is dextrorotatory in nature.
Reason: ‘D’ represents its dextrorotatory nature.
विकल्प
Assertion and reason both are correct statements and reason explains the assertion.
Both assertion and reason are wrong statements.
Assertion is correct statement and reason is wrong statement.
Assertion is wrong statement and reason is correct statement.
Assertion and reason both are correct statements but reason does not explain assertion.
उत्तर
Assertion is correct statement and reason is wrong. statement.
Explanation:
Glucose is correctly named as D(+)-glucose. ‘D’ before the name of glucose represents the configuration whereas ‘(+)’ represents dextrorotatory nature of the molecule. The letters ‘D’ or ‘L’ before the name of any compound indicate the relative configuration of a particular stereoisomer.
APPEARS IN
संबंधित प्रश्न
Write the product when D-glucose reacts with conc. HNO3.
Write the reactions involved when D-glucose is treated with the following reagent:
Br2 water
The symbols D and L represents ____________.
Choose the correct relationship for glucose and fructose:
Why does compound (A) given below not form an oxime?
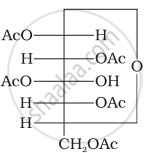
(A)
How will you distinguish 1° and 2° hydroxyl groups present in glucose? Explain with reactions.
Write the reactions of D-glucose which can’t be explained by its open-chain structure. How can cyclic structure of glucose explain these reactions?
On the basis of which evidences D-glucose was assigned the following structure?
\[\begin{array}{cc}
\ce{CHO}\\
|\phantom{....}\\
\phantom{..}\ce{(CHOH)4}\\
|\phantom{....}\\
\phantom{..}\ce{CH2OH}
\end{array}\]
The number of asymmetric carbon atoms in the glucose molecule in open and cyclic form is ______.
When D-glucose reacts with HI, it forms ______.
