Advertisements
Advertisements
प्रश्न
Why does compound (A) given below not form an oxime?
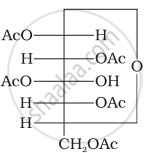
(A)
उत्तर
Glucose pentaacetate (structure A) does not have a free –OH group at C, and therefore, cannot be converted to the open chain form to give a free –CHO group and hence it does not form the oxime.
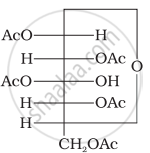
(Structure A)
APPEARS IN
संबंधित प्रश्न
Draw the simple Fisher projection formulae of D - (+) - glucose and D - (-) - fructose
Write the product when D-glucose reacts with conc. HNO3.
Answer the following question.
What is the basic structural difference between glucose and fructose?
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
Glucose does not give Schiff’s test because of the formation of cyclic ____________.
Glucose reacts with acetic anhydride to form ______.
The reaction of glucose with red P + HI is called ____________.
Account for the following:
There are 5 OH groups in glucose
Account for the following:
What happens when D – glucose is treated with the following reagents
HNO3
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
