Advertisements
Advertisements
प्रश्न
Write the reactions involved when D-glucose is treated with the following reagent:
H2N-OH
उत्तर
\[\begin{array}{cc}
\phantom{............}\ce{CHO}\phantom{.............}\ce{CO=N-OH}\\
\phantom{..}|\phantom{..................}|\\
\phantom{........}\ce{(CHOH)4->[NH2OH](CHOH)4}\\
\phantom{..}|\phantom{..................}|\\
\phantom{.........}\ce{\underset{\text{D-glucose}}{CH2OH}}\phantom{..........}\ce{\underset{\text{Oxime}}{CH2OH}}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write the reaction that indicates the presence of -CHO group in glucose
Enumerate the reactions of D-glucose which cannot be explained by its open chain structure.
Write the product when D-glucose reacts with conc. HNO3.
Which of the following statements is incorrect regarding glucose?
Which is the least stable form of glucose?
Which of the following pairs represents anomers?
Why does compound (A) given below not form an oxime?
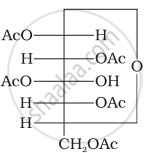
(A)
When D-glucose reacts with HI, it forms ______.
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
Give a reason for the following observations:
Penta-acetate of glucose does not react with hydroxylamine.
