Advertisements
Advertisements
प्रश्न
Draw the structure of boric acid showing hydrogen bonding. Which species is present in water? What is the hybridisation of boron in this species?
उत्तर
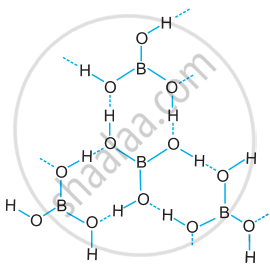
Structure of boric acid; the dotted lines represent hydrogen bonds
[B(OH)4]– units are present in water. Boron has sp3 hybridisation in [B(OH)4]– unit.
APPEARS IN
संबंधित प्रश्न
Is boric acid a protic acid? Explain.
Explain what happens when boric acid is heated.
Boric acid is polymeric due to ______.
Boric acid is an acid because its molecule ______.
What are boranes? Give chemical equation for the preparation of diborane.
Which of the following is a FALSE statement about boric acid, \[\ce{H3BO3}\]?
Boric acid heated to red hot gives ______.
Which of the following compounds are formed when boron trichloride is treated with water?
Boron reacts with nitric acid to form ______.
Which one of the following methods is used to prepare borax crystals?
