Advertisements
Advertisements
प्रश्न
Dry HCl gas does not change the colour of dry blue litmus paper. Why?
Why does dry HCl gas not change the colour of the dry litmus paper?
उत्तर
The only thing that affects a substance's pH are its ions. In the presence of water, HCl produces hydrogen ions. In the absence of water, the H+ ion and HCl molecules cannot be separated. Because dry HCI lacks H+ ions, it does not exhibit the characteristics of an acid.
APPEARS IN
संबंधित प्रश्न
From which plant is litmus paper or litmus solution obtained?
- Moss
- Rose
- Hibiscus
- Lichen
A solution reacts with crushed egg-shells to give a gas that turns lime-water milky. The solution contains ______.
Write a word equation and then a balanced equation for the reaction taking place when:
Dilute hydrochloric acid reacts with magnesium ribbon.
Name one natural source of the following acids:
Lactic acid
A substance X which is used as an antacid reacts with dilute hydrochloric acid to produce a gas Y which is used in one type of fire-extinguisher. Name the substance X and gas Y. Write a balanced equation for the chemical reaction which takes place.
A first-aid manual suggests that vinegar should be used to treat wasp stings and baking soda for bee stings. What does this information tell you about the chemical nature of:
wasp stings?
What is the chemical formula of baking soda?
What happens when a solution of sodium hydrogencarbonate is heated? Write equation of the reaction involved.
State two uses each of the following compounds:
Hydrochloric acid
Write the chemical formula of washing soda. How can it be obtained from baking soda? List two industries in which washing soda is used for other purposes than washing clothes.
Acids turn blue litmus paper to ______.
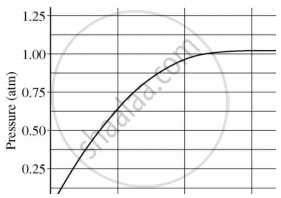
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
Which of the following gives the correct increasing order of acidic strength?
What happens when nitric acid is added to egg shell?
Which acid is present in milk?
______ & ______ metals do not react with HCl or HNO3.
The taste of acid is ______.
On placing a copper coin in a test tube containing green ferrous sulphate solution, it will be observed that the ferrous sulphate solution ______.
The metal that will not produce hydrogen gas when reacted with dilute acids.
