Advertisements
Advertisements
प्रश्न
EΘ values of some redox couples are given below. On the basis of these values.
EΘ values: \[\ce{Br2 /Br- = + 1.90; Ag+ / Ag(s) = + 0.80}\];
\[\ce{Cu^{2+} / Cu(s) = + 0.34; I2(s) / I- = 0.54}\]
विकल्प
\[\ce{Cu}\] will reduce \[\ce{Br-}\]
\[\ce{Cu}\] will reduce \[\ce{Ag}\]
\[\ce{Cu}\] will reduce \[\ce{I-}\]
\[\ce{Cu}\] will reduce \[\ce{Br2}\]
उत्तर
\[\ce{Cu}\] will reduce \[\ce{Br2}\]
Explanation:
Reduction potential is defined as the tendency of the specie to get reduced. More positive the value of EΘ, greater is the tendency of the species to get reduced and stronger is the oxidising agent.
On the basis of the given EΘ values, the order of getting reduced is:
\[\ce{Br2Ag+I2Cu^{2+}}\]
Hence, \[\ce{Cu}\] has the least tendency to get reduced and will itself gets oxidise and reduce other species as: \[\ce{Br2, Ag+}\] and \[\ce{I2}\].
APPEARS IN
संबंधित प्रश्न
Write the formulae for the following compound:
Mercury(II) chloride
According to the classical concept, involves one of the following?
(A) \[\ce{HOCl + H2O2 -> H3O^+ + Cl^- + O2}\]
(B) \[\ce{I2 + H2O2 + 2OH^- -> 2I^- + 2H2O + O2}\]
Choose the correct option.
Given below are two statements:
Statement I: The E° value for `("Ce"^(4+))/("Ce"^(3+))` is +1.74 V.
Statement II: Ce is more stable in Ce4+ state than Ce3+ state.
In the light of the above statements, choose the correct answer from the options given below:
In which of the following reaction, H2O2 is behaving as a reducing agent:
Ge (II) compounds are powerful reducing agents, whereas Pb (IV) compounds are strong oxidants. It can be due to ______.
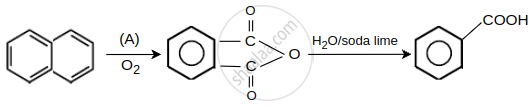
Oxidizing agent (A) used is ______.
The compound that cannot act both as oxidizing and reducing agent is ______.
Copper becomes green when exposed to moist air for a long period. This is due to ______
Which one of the following cannot function as an oxidizing agent?
