Advertisements
Advertisements
प्रश्न
Elements A, B and C have atomic numbers 9, 20 and 10 respectively.
- State which one is
(1) a non-metal,
(2) a metal,
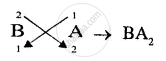
(3) chemically inert.[A,B,C] - Write down the formula of the compound formed by two of the above elements.[BA2]
उत्तर
- 9A 20B 10C
Atomic number of:
A = 9 = number of electrons = 2, 7
B = 20 = number of electons = 2, 8, 8, 2
C = 10 = number of electrons = 2, 8
(1) A non-metal = A
(2) A metal = B
(3) Chemically inert = C - Compound formed is A1−, B2+
APPEARS IN
संबंधित प्रश्न
Fill in the blanks.
Isotopes are the atoms of ______ element having the ______ atomic number but ______ mass number.
The atom of an element is made up of 4 protons, 5 neutrons and 4 electrons. What is its atomic number and mass number?
FILL IN THE BLANK
Atomic theory of matter was given by ..........................
(a) What is meant by (i) atomic number, and (ii) mass number, of an element? Explain with the help of an example.
(b) What is the relation between the atomic number and mass number of an element?
(c) If an element M has mass number 24 and atomic number 12, how many neutrons does its atom contain?
An atom of an element has two electrons in the M shell.
What is the (a) atomic number (b) number of protons in this element?
Define the term ‘mass number’ of an atom.
If an atom ‘A’ has atomic number 19 and mass number 39, state –
- Its electronic configuration.
- The number of valence electrons it possesses.
The diagram represents an isotope of hydrogen [H]. Answer the following:

Atomic no. = 1
Mass no. = 1
Are isotopes atoms of the same element or different elements.
The ratio of the radii of hydrogen atom and its nucleus is ~ 105. Assuming the atom and the nucleus to be spherical,
- what will be the ratio of their sizes?
- If atom is represented by planet earth ‘Re ’ = 6.4 ×106 m, estimate the size of the nucleus.
Give short answer:
what is the atomic mass number? mention its formula.
