Advertisements
Advertisements
प्रश्न
Esters are sweet-smelling substances and are used in making perfumes. Describe an activity for the preparation of an ester with the help of a well labelled diagram. Write an equation for the chemical reaction involved in the formation of the ester. Also write the names of all the substances involved in the process of esterification.
उत्तर
Esters are sweet-smelling substances which are used to make perfumes. The process in which a carboxylic acid reacts with an alcohol to form an ester is called esterification.
An ester is formed when ethanol (alcohol) reacts with ethanoic acid, on heating, in the presence of a few drops of concentrated sulphuric acid. The name of the ester formed is ethyl ethanoate.
Let's prepare an ester.
(1) In a test tube, take 1 ml of ethanoic acid and add some ethanol to it along with a few drops of concentrated sulphuric acid.
(2) Warm the test tube in a beaker, containing hot water, for 5 minutes.
(3) Take about 50 ml of water in another beaker and pour the contents of the test tube into it. When you smell it, a sweet fragrance is obtained indicating the formation of an ester.
The diagrammatic representation of the above process is as follows:

The chemical equation involved in the formation of ester is as follows:
`CH_3 COOH + C_2 H_5 OH` 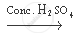 `CH_3 COOC_2 H_5 + H_2 O`
`CH_3 COOC_2 H_5 + H_2 O`
APPEARS IN
संबंधित प्रश्न
In an experiment to study the properties of ethanoic acid, a student takes about 3 mL of ethanoic acid in a dry test tube. He adds an equal amount of distilled water to it and shakes the test tube well. After some time he is likely to observe that
(A) a colloid is formed in the test tube.
(B) the ethanoic acid dissolves readily in water.
(C) the solution becomes light orange.
(D) water floats over the surface of ethanoic acid.
Complete the following chemical equations:
C2H5OH+CH3COOH`("conc."H_2SO_4)/`>
State any two uses of esters.
Which of the following observations is true about dilute solution of acetic acid?
(A) It smells like vinegar and turns red litmus blue
(B) It smells like onion and turns blue litmus red
(C) It smells like orange and turns red litmus blue
(D) It smells like vinegar and turns blue litmus red
Why is Soda lime used not only NaOH?
Name the first four members of alphatic carboxylic acid.
Why is pure acetic acid also known as glacial acetic acid?
A student while observing the properties of acetic acid would report that this smells like ______.
When ethanoic acid is treated with NaHCO^ the gas evolved is ______.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
