Advertisements
Advertisements
Question
Esters are sweet-smelling substances and are used in making perfumes. Describe an activity for the preparation of an ester with the help of a well labelled diagram. Write an equation for the chemical reaction involved in the formation of the ester. Also write the names of all the substances involved in the process of esterification.
Solution
Esters are sweet-smelling substances which are used to make perfumes. The process in which a carboxylic acid reacts with an alcohol to form an ester is called esterification.
An ester is formed when ethanol (alcohol) reacts with ethanoic acid, on heating, in the presence of a few drops of concentrated sulphuric acid. The name of the ester formed is ethyl ethanoate.
Let's prepare an ester.
(1) In a test tube, take 1 ml of ethanoic acid and add some ethanol to it along with a few drops of concentrated sulphuric acid.
(2) Warm the test tube in a beaker, containing hot water, for 5 minutes.
(3) Take about 50 ml of water in another beaker and pour the contents of the test tube into it. When you smell it, a sweet fragrance is obtained indicating the formation of an ester.
The diagrammatic representation of the above process is as follows:

The chemical equation involved in the formation of ester is as follows:
`CH_3 COOH + C_2 H_5 OH` 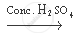 `CH_3 COOC_2 H_5 + H_2 O`
`CH_3 COOC_2 H_5 + H_2 O`
APPEARS IN
RELATED QUESTIONS
A student puts a drop of reaction mixture of a saponification reaction first a blue litmus paper and then on a red litmus paper. He may observe that:
(a) There is no change in the blue litmus paper and the red litmus paper turns white.
(b) There is no change in the red litmus paper and the blue litmus paper turns red.
(c) There is no change in the blue litmus paper and the red litmus paper turns blue.
(d) No change in colour is observed in both the litmus papers
When zinc powder is added to acetic acid ______________
(a) the mixture becomes warm
(b) a gas is evolved
(c) the colour of the mixture becomes yellow
(d) a solid settles at the bottom
What do you notice when acetic acid reacts with litmus?
A student is studying the properties of acetic acid. List two physical properties of acetic acid he observes. What happens when he adds a pinch of sodium hydrogen carbonate to this acid? Write any two observations.
Two statements are given - one labelled Assertion (A) and the other labelled Reason (R).
Assertion (A): Esterification is a process in which a sweet-smelling substance is produced.
Reason (R): When esters react with sodium hydroxide an alcohol and sodium salt of carboxylic acid are obtained.
Ketone : –CO– : : Ester : _______
Name the oxidising agent used in the conversion of ethanol to ethanoic acid.
Observe the diagram given below and answer the questions:
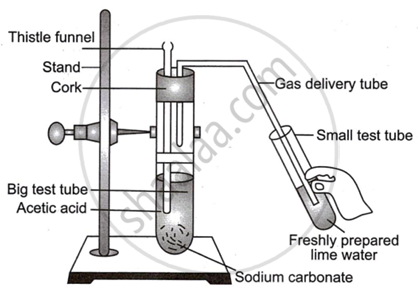
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
