Advertisements
Advertisements
प्रश्न
Ethane burns in oxygen to form CO2 and H2O according to the equation:
`2C_2H_6+7O_2 -> 4CO_2 + 6H_2O`
If 1250 cc of oxygen is burnt with 300 cc of ethane.
Calculate:
1) the volume of `CO_2` formed
2) the volume of unused `O_2`
उत्तर
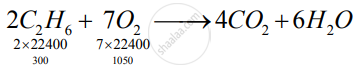
So, ethane is limiting reagent.
1) 2 x 22400 cc ethane give -> 4 x 22400 cc `CO_2`
∴ 300 cc ethane given → `(4xx22400xx300)/(2xx22400) "cc" Co_2`
= 600 cc CO2
2) For 300 cc Ethane 1050 cc of O2 will be required.
So unused O2 is (1250-1050) = 200 cc
APPEARS IN
संबंधित प्रश्न
Identify the term or substance based on the descriptions given below:
The property by virtue of which the compound has the same molecular formula but different structural formulae.
A compound contains 87.5% by mass of nitrogen and 12.5% by mass of hydrogen. Determine the empirical formula of this compound.
The compound A has the following percentage composition by mass: C =26.7%, O = 71.1%, H = 2.2%.
Determine the empirical formula of A.(Answer to one decimal place) (H=1,C=12,O=16)
Determine the empirical formula of the compound whose composition by mass is 42% nitrogen, 48% oxygen and 9% hydrogen.[N=14,O=16,H=1]
The percentage composition of sodium phosphate, as determined by analysis is : 42.1% Na, 18.9% P, 39% of O. Find the empirical formula of the compound.
[H =1, N =14, Na = 23, P = 31, Cl = 35.5, Pt = 195]
A compound 'X' consists of 4.8% of C and 95.2% of Br by mass.
Determine the empirical formula of this compound.
[C = 12, Br = 80]
Give the empirical formula of C6H18O3
Silicon (Si = 28) forms a compound with chlorine (Cl = 35.5) in which 5.6 g of silicon combines with 21.3 g of chlorine. Calculate the empirical formula of the compound.
A compound has the following percentage composition by mass: carbon 14.4%, hydrogen 1.2% and chlorine 84.5%. Determine the empirical formula of this compound. Work correctly to 1 decimal place. (H = 1; \[\ce{C}\] = 12; \[\ce{Cl}\] = 35.5)
Pratik heated 11.2 grams of element ‘M’ (atomic weight 56) with 4.8 grams of element 'N' (atomic weight 16) to form a compound. Find the empirical formula of the compound obtained by Pratik.
