Advertisements
Advertisements
प्रश्न
Ethane burns in oxygen to form CO2 and H2O according to the equation:
`2C_2H_6+7O_2 -> 4CO_2 + 6H_2O`
If 1250 cc of oxygen is burnt with 300 cc of ethane.
Calculate:
1) the volume of `CO_2` formed
2) the volume of unused `O_2`
उत्तर
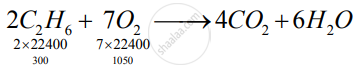
So, ethane is limiting reagent.
1) 2 x 22400 cc ethane give -> 4 x 22400 cc `CO_2`
∴ 300 cc ethane given → `(4xx22400xx300)/(2xx22400) "cc" Co_2`
= 600 cc CO2
2) For 300 cc Ethane 1050 cc of O2 will be required.
So unused O2 is (1250-1050) = 200 cc
APPEARS IN
संबंधित प्रश्न
Consider the following reaction and based on the reaction answer the questions that follow:

Calculate:
1) the quantity in moles of (NH4)2Cr2O7 if 63gm of(NH4)2Cr2O7 is heated.
2) the quantity in moles of nitrogen formed.
3) the volume in liters or dm3 of N2 evolved at S.T.P.
4) the mass in grams of Cr2O3 formed at the same time
(Atomic masses: H=1, Cr= 52, N=14]
A compound made up of two elements X and Y has an empirical formula X2Y. If the atomic weight of X is 10 and that of Y is 5 and the compound ha a vapor density 25, find its molecular formula.
Find the empirical formula and the molecular formula of an organic compound from the data given below :
C = 75.92%, H = 6.32% and N = 17.76%
The vapour density of the compound is 39.5.
[C = 12, H = 1, N = 14]
Give the empirical formula of C6H6.
Give the empirical formula of: C6H12O6
Give the empirical formula of C2H2
The compound A has the following percentage composition by mass: C =26.7%, O = 71.1%, H = 2.2%.
Determine the empirical formula of A.(Answer to one decimal place) (H=1,C=12,O=16)
Give the empirical formula of C6H18O3
The Empirical formula of an organic compound is CHCl2.
If its relative molecular mass is 168, what is its molecular formula?
[At. Wt. C = 12, H = 1, Cl = 35.5]
Calculate:
Write the empirical formula of C8H18.
