Advertisements
Advertisements
प्रश्न
Ethanoic acid reacts with the basic salt, sodium carbonate, to form a salt, named ______, water and carbon dioxide gas.
विकल्प
sodium ethanoate
ethyl ethanoate
ethanol
ethyl sodium
उत्तर
Ethanoic acid reacts with the basic salt, sodium carbonate, to form a salt, named sodium ethanoate, water and carbon dioxide gas.
APPEARS IN
संबंधित प्रश्न
What are esters ?
Fill in the blanks from the choices given within brackets:
The basicity of acetic acid is-------- (3, 1, 4).
Give a reason for Conductivity of dilute hydrochloric acid is greater than that of acetic acid
propanol into propanoic acid?
Name the process in each case and write the equations of the reactions involved.
Name the product formed and give an appropriate chemical equation for the following:
Ethanol oxidised by acidified potassium dichromate?
Complete:
The next higher homologue of ethanoic acid is ______.
Name the substance used to change acetic acid to acetic anhydride?
Give two tests to show that CH3COOH is acidic in nature ?
While studying saponification reactions, the following comments were noted down by the students :
(I) Soap is a salt of fatty acids.
(II) The reaction mixture is basic in nature.
(III) In this reaction heat is absorbed.
(IV) This reaction is not a neutralisation reaction.
Which of these are the correct comments ?
(A) I and III only
(B) I, II and III
(C) II, III and IV
(D) I and II only
A student takes 2 mL acetic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He makes the following observations:
I. A colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are:
(A) I, II, and III
(B) II, III and IV
(C) III, IV and I
(D) IV, I and II
On adding acetic acid to sodium hydrogen carbonate in a test tube, a student observes
(A) no reaction
(B) a colourless gas with pungent smell
(C) bubbles of a colourless and odourless gas
(D) a strong smell of vinegar
Write the molecular formulae and structure formula of ethanic acid?
Ethanoic acid _________.
Explain the following reaction with an example.
Esterification
Ethanoic acid turns ______ litmus to ______
A few drops of ethanoic acid were added to solid sodium carbonate. The observation made was that ______.
Ester is formed by the reaction between ______.
Write the chemical equation for the ethanol to ethanoic acid of an oxidation reaction.
Observe the diagram given below and answer the questions:
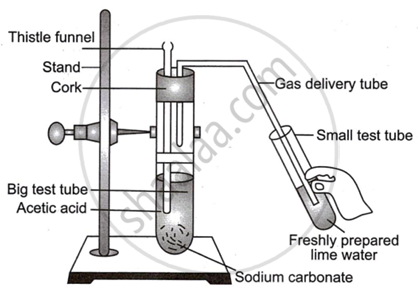
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
