Advertisements
Advertisements
प्रश्न
Observe the diagram given below and answer the questions:
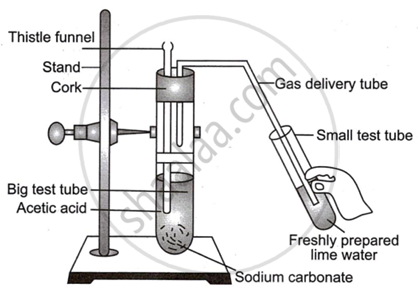
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
उत्तर
- The reactants are acetic acid \[\ce{(CH3COOH)}\] and sodium carbonate \[\ce{(Na2CO3)}\].
- The gas produced is carbon dioxide \[\ce{(CO2)}\].
- When carbon dioxide gas is passed through lime water, it turns lime water milky or cloudy due to the formation of calcium carbonate. Therefore, the colour change in the lime water would be from clear to milky.
- In the above experiment, instead of sodium carbonate, sodium bicarbonate can be used to get the same products.
- Acetic acid is widely used in the culinary arts as a key ingredient in vinegar. It is also used industrially as a chemical reagent for the production of chemical compounds, in the manufacture of plastics, and in the food industry as a preservative and flavouring agent.
APPEARS IN
संबंधित प्रश्न
A student adds a few drops of ethanoic acid to test tubes X, Y and Z containing aqueous solutions of sodium chloride, sodium hydroxide and sodium carbonate, respectively. If he now brings a burning splinter near the mouth of the test tubes immediately after adding ethanoic acid in each one of them, in which of the test tube or test tubes the flame will be extinguished?
(A) X and Y
(B) Y and Z
(C) X and Z
(D) only Z
Fill in the blanks from the choices given within brackets:
The basicity of acetic acid is-------- (3, 1, 4).
What is the common name of ethanoic acid?
Write the formulae of methanoic acid.
Three organic compounds A, B and C have the following molecular formulae: C4H10O
Which compound contains a carboxyl group? Write its name and structural formula.
Why is the conversion of ethanol into ethanoic acid an oxidation reaction?
Name two oxidising agents which can oxidise ethanol to ethanoic acid.
How does ethanoic acid react with sodium hydrogen carbonate? Give equation of the reaction which takes place.
When ethanoic acid reacts with sodium hydrogen carbonate, then a salt X is formed and a gas Y is evolved. Name the salt X and gas. Y Describe an activity with the help of a labelled diagram of the apparatus used to prove that the evolved gas is the one which you have named. Also write the chemical equation of the reaction involved.
The substance which can produce brisk effervescence with baking soda solution is:
(a) ethanol
(b) vegetable oil
(c) vinegar
(d) soap solution(b) hydrocarbon ends directed towards the centre and ionic ends directed outwards
In a soap micelle, the soap molecules are arranged radially with the hydrocarbon ends, i.e. hydrophobic, directed towards the centre; and, ionic ends, i.e. hydrophilic, directed outwards.
What is glacial acetic acid ?
Complete:
The next higher homologue of ethanoic acid is ______.
How is acetic acid prepared from acetylene?
Acetic acid is a typical acid. Write one equation in case of its reactions with a carbonate.
On adding NaHCO3 to acetic acid, a gas is evolved which turns lime water milky due to the formation of:
(1) Calcium bicarbonate
(2) Calcium hydroxide
(3) Calcium carbonate
(4) Calcium acetate
What is the action of acetic acid with litmus paper?
How will you carry out the following conversions?
Ethene to acetic acid
Draw the structure formula of ethyne.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
