Advertisements
Advertisements
प्रश्न
Observe the diagram given below and answer the questions:
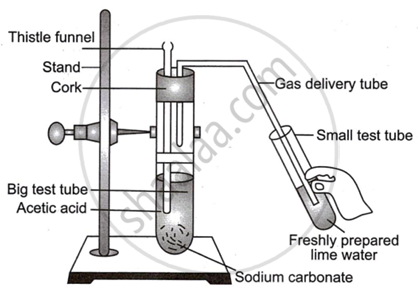
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
उत्तर
- The reactants are acetic acid \[\ce{(CH3COOH)}\] and sodium carbonate \[\ce{(Na2CO3)}\].
- The gas produced is carbon dioxide \[\ce{(CO2)}\].
- When carbon dioxide gas is passed through lime water, it turns lime water milky or cloudy due to the formation of calcium carbonate. Therefore, the colour change in the lime water would be from clear to milky.
- In the above experiment, instead of sodium carbonate, sodium bicarbonate can be used to get the same products.
- Acetic acid is widely used in the culinary arts as a key ingredient in vinegar. It is also used industrially as a chemical reagent for the production of chemical compounds, in the manufacture of plastics, and in the food industry as a preservative and flavouring agent.
APPEARS IN
संबंधित प्रश्न
Complete the following chemical equations:CH3COOC2H5+NaOH→
Complete the following chemical equations: CH3COOH+NaHCO3→
Give the common names and IUPAC names of the following compounds of CH3COOH.
What will you observe when dilute ethanoic acid and dilute hydrochloric acid are put on universal indicator paper, one by one? What does it show?
What type to compound is CH3COOH?
An organic compound X of molecular formula C2H4O2 gives brisk effervescence with sodium hydrogen carbonate. Give the name and formula of X.
When ethanoic acid reacts with sodium hydrogen carbonate, then a salt X is formed and a gas Y is evolved. Name the salt X and gas. Y Describe an activity with the help of a labelled diagram of the apparatus used to prove that the evolved gas is the one which you have named. Also write the chemical equation of the reaction involved.
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is added to alcohol to denature it?
Name the product formed and give an appropriate chemical equation for the following:
Ethanol oxidised by acidified potassium dichromate?
Give the structural formulae of acetic acid.
What is glacial acetic acid ?
A student adds 4 mL of acetic acid to a test tube containing 4 mL of distilled water. He then shakes the test tube and leaves it to settle. After about 10 minutes he observes:
(A) a layer of water over the layer of acetic acid
(B) a layer of acetic acid over the layer of water
(C) a precipitate settling at the bottom of the test tube
(D) a clear colourless solution
Using appropriate catalysts, ethane can be oxidized to an alcohol. Name the alcohol formed when ethane is oxidized.
Choose the correct answer:
Select the acid which contains four hydrozen atoms in it.
State how the following conversions can be carried out:
Ethyl chloride to Ethene.
Choose the correct option from given alternative:
When sodium hydrogen carbonate is added to ethanoic acid a gas evolves. Consider the following statements about the gas evolved?
(A) It turns lime water milky.
(B) It is evolved with brisk effervescence.
(C) It has a smell of burning sulfur.
(D) It is also a by-product of respiration.
Which of the following represents saponification reaction?
Ethanoic acid reacts with the basic salt, sodium carbonate, to form a salt, named ______, water and carbon dioxide gas.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
Give the balanced chemical equation of the following reaction:
Neutralization of NaOH with ethanoic acid.
