Advertisements
Advertisements
प्रश्न
Explain graphically Freundlich adsorption isotherm.
उत्तर
Graphical representation of the Freundlich adsorption isotherm: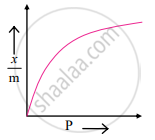
Variation of x/m with P according to Freundlich equation (Adsorption isotherm)
- Freundlich proposed the following empirical equation for the adsorption of a gas on a solid.
`"x"/"m" = "k P"^(1//"n")` (n > 1) ....(i)
where.
x = Mass of the gas adsorbed
m = Mass of the adsorbent
`"x"/"m"` = Mass of gas adsorbed per unit mass of adsorbent
P = Equilibrium pressure
k and n are constants which depend on the nature of adsorbate, adsorbent and temperature. - The graphical representation of the Freundlich equation is as shown in the adjacent plot of `x/m` vs ‘P’.
- In case of solution, P in the equation (i) is replaced by the concentration (C) and thus,
`"x"/"m" = "k C"^(1//"n")` ....(ii)
- By taking logarithm on both sides of the equation (ii), we get
`log "x"/"m" = log "k" + 1/"n" log "C"` ...(iii) - On plotting a graph of log `"x"/"m"` against log C or log P, a straight line is obtained as shown in the adjacent plot. The slope of the straight line is `1/"n"` and intercept on Y-axis is log k.
- The factor `1/"n"` ranges from 0 to 1. Equation (iii) holds good over a limited range of pressures.
a. When `1/"n"-> 0, "x"/"m" ->` constant, the adsorption is independent of pressure.
b. When `1/"n" = 1, "x"/"m" = "kP", i.e., "x"/"m" prop "P"`, the adsorption varies directly with pressure.
c. The experimental isotherms tend to saturate at high pressure.
APPEARS IN
संबंधित प्रश्न
What is an adsorption isotherm?
Describe Freundlich adsorption isotherm.
What do you understand by activation of adsorbent? How is it achieved?
In reference to Freundlich adsorption isotherm, write the expression for adsorption of gases on solids in the form of an equation.
Answer in one sentence:
Write an equation for Freundlich adsorption isotherm.
The slope and intercept for plot of `log_10 "x"/"m"` against log10P for Freundlich equation are ____________ and ____________ respectively.
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The plot of log `"x"/"m"`· versus log P is shown in the given m graph. `"x"/"m"` is proportional to
Freundlich's equation for adsorption of gas on solid is represented as ____________.
Which of the following curves is in accordance with Freundlich adsorption isotherm?
Freundlich adsorption isotherm is given by the expression `x/m = kp^(1/n)` which of the following conclusions can be drawn from this expression.
(i) When `1/n` = 0, the adsorption is independent of pressure.
(ii) When `1/n` = 0, the adsorption is directly proportional to pressure.
(iii) When n = 0, `x/m` vs p graph is a line parallel to x-axis.
(iv) When n = 0, plot of `x/m` vs p is a curve.
Adsorption of gases on the solid surface is generally exothermic because.
Langmuir adsorption isotherm works particularly well.
In the freundilch adsorption isotherm, the value of x/m is 0.4 under a pressure of 0.2 at m calculate the value of the interact
Which of the following statement is INCORRECT regarding the adsorption of a gas on the surface of the solid?
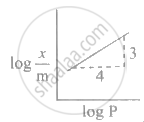
In Freundlich adsorption isotherm, slope of AB line is:
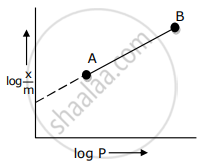
Adsorption of a gas follows Freundlich adsorption isotherm. x is the mass of the gas adsorbed on mass m of the adsorbent. The x plot of log `x/m` versus log p is shown in the given graph. `x/m` is proportional to ______.
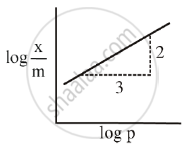
For Freundlich adsorption isotherm, a plot of log (x/m) (Y-axis) and log p (x-axis) gives a straight line. The intercept and slope for the line is 0.4771 and 2, respectively. The mass of gas, adsorbed per gram of adsorbent if the initial pressure is 0.04 atm, is ______ × 10-4 g. (log 3 = 0.4771)
