Advertisements
Advertisements
प्रश्न
Explain how, mercury is extracted from its sulphide ore (cinnabar). Give equations of the reactions involved.
उत्तर
Cinnabar is a sulphide ore of mercury. The name of the compound present in the cinnabar ore is mercury (II) sulphide. Its chemical formula is HgS. Mercury is a less reactive metal and it can be extracted from its sulphide ore cinnabar by heating it in air alone.
1. It is first heated in the presence of surplus air (roasting process). This converts mercury (II) sulphide to mercury (II) oxide.
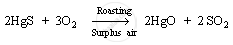
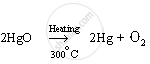
Mercury (II) oxide, so formed, is heated to around 300°C and it gets reduced to mercury metal.
APPEARS IN
संबंधित प्रश्न
Name two metals which are always found in combined state.
Calamine ore can be converted into zinc oxide by the process of:
(a) dehydration
(b) roasting
(c) calcination
(d) sulphonation
Name the following:
The substance added to get rid of gangue in the extraction of metal.
Define the term : Slag
Zinc sulphide : Roasting : : zinc carbonate : _______
Explain Bayer’s process.
The process in which a carbonate ore is heated strongly in the absence of air to convert it into metal oxide is called ____________.
Which of the statements about the reaction, \[\ce{ZnO + CO -> Zn + CO2}\] is correct?
Iqbal treated a lustrous, divalent element M with sodium hydroxide. He observed the formation of bubbles in reaction mixture. He made the same observations when this element was treated with hydrochloric acid. Suggest how can he identify the produced gas. Write chemical equations for both the reactions.
A mineral from which the metal can be extracted economically and conveniently is known as ______.
