Advertisements
Advertisements
प्रश्न
Explain how, mercury is extracted from its sulphide ore (cinnabar). Give equations of the reactions involved.
उत्तर
Cinnabar is a sulphide ore of mercury. The name of the compound present in the cinnabar ore is mercury (II) sulphide. Its chemical formula is HgS. Mercury is a less reactive metal and it can be extracted from its sulphide ore cinnabar by heating it in air alone.
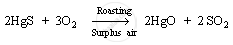
1. It is first heated in the presence of surplus air (roasting process). This converts mercury (II) sulphide to mercury (II) oxide.
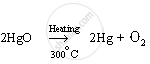
Mercury (II) oxide, so formed, is heated to around 300°C and it gets reduced to mercury metal.
APPEARS IN
संबंधित प्रश्न
In the extraction of aluminium: Why is it necessary to replace anodes time to time?
A zinc ore gave CO2 on treatment with a dilute acid. Identify the ore and write its chemical formula.
Name one metal which is extracted by reduction with carbon.
How is zinc extracted from its carbonate or (calamine)? Explain with equations.
Rock salt is an ore of one of the following metals. This metal is:
(a) Mn
(b) Na
(c) Fe
(d) Cu
Name the methods by which concentrated ore is converted to metallic oxide.
Answer the following question:
Carbon cannot reduce the oxides of sodium, magnesium, and aluminum to their respective metals. Why? Where are these metals placed in the reactivity series? How are these metals obtained from their ores? Take an example to explain the process of extraction along with chemical equations.
In the electrolytic reduction of alumina, the lining of graphite acts as an anode.
Identify who I am!
Ore of Aluminum- ______
In thermite welding a mixture of ____________ is ignited with a burning magnesium ribbon which produces molten iron metal as a large amount of heat is evolved.
