Advertisements
Advertisements
प्रश्न
Explain in detail the Carnot heat engine.
उत्तर
In the year 1824, a young French engineer Sadi Carnot proved that a certain reversible engine operated in a cycle between hot and cold reservoirs can have maximum efficiency. This engine is called the Carnot engine.
A reversible heat engine operating in a cycle between two temperatures in a particular way is called a Carnot Engine. The Carnot engine has four parts which are given below.
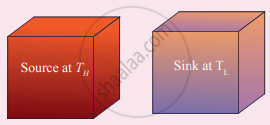
(i) Source: It is the source of heat maintained at constant high-temperature TH. Any amount of heat can be extracted from it, without changing its temperature.
Carnot engine
(ii) Sink: It is a cold body maintained at a constant low-temperature TL. It can absorb any amount of heat.
(iii) Insulating stand: It is made of perfectly non-conducting material. Heat is not conducted through this stand.
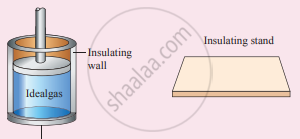
(iv) Working substance: It is an ideal gas enclosed in a cylinder with perfectly non-conducting walls and a perfectly conducting bottom. A non-conducting and frictionless piston is fitted in it.
APPEARS IN
संबंधित प्रश्न
Draw a p-V diagram and explain the concept of positive and negative work. Give one example each.
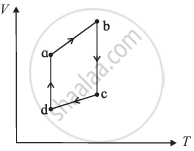
The figure shows the V-T diagram for one cycle of a hypothetical heat engine which uses the ideal gas. Draw the p-V diagram diagram of the system.
State Kelvin-Planck's statement of the second law of thermodynamics.
What are the processes involves in a Carnot engine?
Explain the heat engine and obtain its efficiency.
Derive the expression for Carnot engine efficiency.
For a heat engine operating between temperatures t1 °C and t2 °C, its efficiency will be ______.
Heat engine transfers ______.
Which statement is incorrect?
Let η1 is the efficiency of an engine at T1 = 447°C and T2 = 147°C while η2, is the efficiency at T1 = 947°C and T2 = 47°C. The ratio `eta_1/eta_2` will be ______.
