Advertisements
Advertisements
Question
Explain in detail the Carnot heat engine.
Solution
In the year 1824, a young French engineer Sadi Carnot proved that a certain reversible engine operated in a cycle between hot and cold reservoirs can have maximum efficiency. This engine is called the Carnot engine.
A reversible heat engine operating in a cycle between two temperatures in a particular way is called a Carnot Engine. The Carnot engine has four parts which are given below.
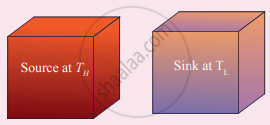
(i) Source: It is the source of heat maintained at constant high-temperature TH. Any amount of heat can be extracted from it, without changing its temperature.
Carnot engine
(ii) Sink: It is a cold body maintained at a constant low-temperature TL. It can absorb any amount of heat.
(iii) Insulating stand: It is made of perfectly non-conducting material. Heat is not conducted through this stand.
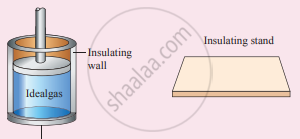
(iv) Working substance: It is an ideal gas enclosed in a cylinder with perfectly non-conducting walls and a perfectly conducting bottom. A non-conducting and frictionless piston is fitted in it.
APPEARS IN
RELATED QUESTIONS
Draw a p-V diagram and explain the concept of positive and negative work. Give one example each.
The efficiency of a heat engine working between the freezing point and boiling point of water is ____________.
State Clausius form of the second law of thermodynamics.
State Kelvin-Planck's statement of the second law of thermodynamics.
Define heat engine.
What are the processes involves in a Carnot engine?
Suppose a person wants to increase the efficiency of the reversible heat engine that is operating between 100°C and 300°C. He had two ways to increase efficiency.
- By decreasing the cold reservoir temperature from 100°C to 50°C and keeping the hot reservoir temperature constant
- by increasing the temperature of the hot reservoir from 300°C to 350°C by keeping the cold reservoir temperature constant.
Which is the suitable method?
A Carnot engine whose efficiency is 45% takes heat from a source maintained at a temperature of 327°C. To have an engine of efficiency of 60% what must be the intake temperature for the same exhaust (sink) temperature?
10 One mole of a van der Waals' gas obeying the equation `("P" + "a"/"V"^2)`(V - b) = RT undergoes the quasi-static cyclic process which is shown in the P-V diagram. The net heat absorbed by the gas in this process is ______
The thermal efficiency of a heat engine is 25%. If in one cycle the heat absorbed from the hot reservoir is 50000 J, what is the heat rejected to the cold reservoir in one cycle?
