Advertisements
Advertisements
प्रश्न
Explain the bond formation if BeCl2 and MgCl2.
उत्तर
Bond formation of BeCl2:
Be = 4;
Electronic configuration of Be atom is = 1s2 2s2

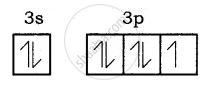

Bond formation of MgCl2:
Mg (Z = 12), 1s2 2s2 2p6 3s2 it loses two of its valence electron and became Mg2+ with inert gas configuration Neon.
The chlorine accepts one electron in its valence shell and because Cl– ion with Ar electron configuration.
Mg + Cl2 → Mg2+ + 2 Cl– → MgCl2
Magnesium cation and two chlorides are attracted by strong electrostatic force to form
MgCl2 crystals.
Mg = 12,
Electronic configuration: 1s2 2s2 2p6 3s2
Mg+2:
Electronic configuration: 1s2 2s2 2p6 3s0
Cl = 17, 1s2 2s2 2p6 3s2 3p5
Cl–:
Electronic configuration: 1s2 2s2 2p6 3s2 3p6


APPEARS IN
संबंधित प्रश्न
In the molecule OA = C = OB, the formal charge on OA, C and OB are respectively.
Draw the Lewis structure for the following species.
O3
What is the Polar Covalent bond?
Draw the Lewis structure for the following species.
NO3–
Draw the Lewis structures for the following species.
\[\ce{NO^-3}\]
Draw the Lewis structure for the following species.
\[\ce{NO^-_3}\]
Draw the Lewis structure for the following species.
SO42-
Draw the Lewis structure for the following species.
`"SO"_4^(2-)`
Draw the Lewis structure for the following species:
\[\ce{NO^-3}\]
Draw the Lewis structure for the following species.
\[\ce{NO^-3}\]
