Advertisements
Advertisements
प्रश्न
Explain the following reaction.
Silicon is heated with methyl chloride at high temperature in the presence of copper.
उत्तर
When silicon reacts with methyl chloride in the presence of copper (catalyst) and at a temperature of about 537 K, a class of organosilicon polymers called methyl-substituted chlorosilanes (MeSiCl3, Me2SiCl2, Me3SiCl, and Me4Si) are formed
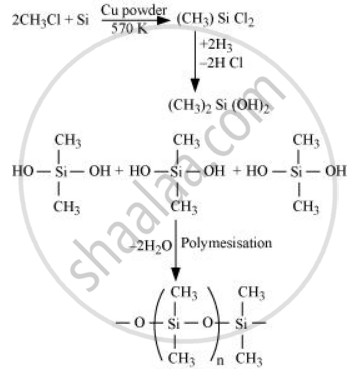
APPEARS IN
संबंधित प्रश्न
Discuss the pattern of variation in the oxidation states of B to Tl.
Discuss the pattern of variation in the oxidation states of C to Pb.
Rationalise the given statement and give a chemical reaction:
Lead (II) chloride reacts with Cl2 to give PbCl4.
Elements of group 14 ________.
Rationalise the given statement and give a chemical reaction:
Lead (IV) chloride is highly unstable towards heat.
Classify the following oxide as neutral, acidic, basic or amphoteric:
CO
Classify the following oxide as neutral, acidic, basic or amphoteric:
B2O3
Write suitable chemical equations to show the nature of the following oxide.
B2O3
Write suitable chemical equations to show the nature of the following oxide.
Al2O3
Explain the following:
Silicon forms \[\ce{SiF^{2-}6}\] ion whereas corresponding fluoro compound of carbon is not known.
Carbon and silicon both belong to the group 14, but inspite of the stoichiometric similarity, the dioxides, (i.e., carbon dioxide and silicon dioxide), differ in their structures. Comment.
Explain the following:
BF3 does not hydrolyse.
Explain the following:
Why does the element silicon, not form a graphite like structure whereas carbon does.
A tetravalent element forms monoxide and dioxide with oxygen. When air is passed over heated element (1273 K), producer gas is obtained. Monoxide of the element is a powerful reducing agent and reduces ferric oxide to iron. Identify the element and write formulas of its monoxide and dioxide. Write chemical equations for the formation of producer gas and reduction of ferric oxide with the monoxide.
Stannous chloride solution when kept in the air turns milky due to the formation of ______.
\[\ce{SiCl4 ->[H2O] (A) ->[\Delta] (B) ->[Na2CO3][heat] (C)}\]. The Compound C is ______.
