Advertisements
Advertisements
प्रश्न
Explain why Calamine solution is applied on the skin when an ant bites.
उत्तर
- Ant injects an acidic liquid (Formic acid) into the skin on biting which causes inflammation, to the skin.
- The acid's effect can be neutralised by rubbing. Calamine solution contains zinc carbonate, which is a very weak base and does not harm the skin.
APPEARS IN
संबंधित प्रश्न
Explain why An antacid tablet is taken when you suffer from acidity.
Explain why factory waste is neutralised before disposing it into the water bodies.
What is a neutralization reaction?
State whether the following statements are true or false. Correct the false statements.
Calamine can be used to treat ant stings.
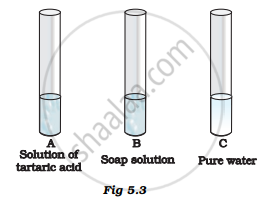
You are provided with three test tubes A, B and C as shown in Figure 5.3 with different liquids. What will you observe when you put
- a piece of blue litmus paper in each test tube.
- a piece of red litmus paper in each test tube.
- a few drops of phenolphthalein solution to each test tube.
Explain two neutralisation reactions related to daily life situations.
When an acid reacts with a base, a neutralization reaction takes place to give salt and water.
Give an important use of neutralization reaction in daily life.
How do treat effluents from the industries?
Visit a doctor. Find out the medicines, he prescribes to treat acidity. Ask him how acidity can be prevented.
