Advertisements
Advertisements
प्रश्न
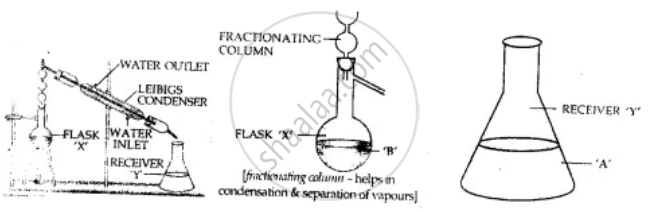
Explain with diagram the process used to – separate the following substance from the given mixtures - Benzene from a mixture of – benezene [b.p. 80°C] & toluene [b.p. 110°C].
उत्तर
By fractional distillation, miscible low boiling point benzene (B.P. 80°C) evaporates on heating the mixture and condenses in and collects in flask ‘Y’ whereas higher boiling pt. Toluene (B.P. 110°C) remains in flask ‘X’ after condensation.
APPEARS IN
संबंधित प्रश्न
Suggest a suitable technique to separate the constituents of the following mixtures. Also give the reason for selecting the particular method.
Salt from sea water
Choose the most appropriate answer
Which of the following property is not shown by compounds?
Tick the most appropriate answer
The methods of separating components of a given mixture are based on the
- physical properties and state of the components.
- colour of the components only.
- state of the components.
- none of these
Name the following:
The compound containing one atom of sulphur and two atoms of oxygen.
State any one method – to separate the following mixtures – A gas-gas mixture containing – two gases with different densities.
Explain with diagram the process used to – separate the following substance from the given mixtures - Lead nitrate [soluble] from a mixture of – lead nitrate & water Le. lead nitrate solution.
Select the correct answer from the choice given below to complete the sentence :
The reactive element from the two monoatomic elements is _____________.
How can you separate a large quantity of tiny bits of paper mixed with pulses/dal?
How will you separate sand and water from their mixture?
A mixture of milk and water can be separated by filtration.
