Advertisements
Advertisements
Question
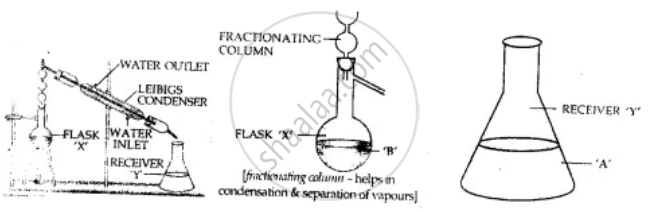
Explain with diagram the process used to – separate the following substance from the given mixtures - Benzene from a mixture of – benezene [b.p. 80°C] & toluene [b.p. 110°C].
Solution
By fractional distillation, miscible low boiling point benzene (B.P. 80°C) evaporates on heating the mixture and condenses in and collects in flask ‘Y’ whereas higher boiling pt. Toluene (B.P. 110°C) remains in flask ‘X’ after condensation.
APPEARS IN
RELATED QUESTIONS
How is sodium chloride different from its constituent elements ?
What is chromatography? For which type of mixture is it used?
Fill in the blan
Iodine, camphor, naphthalene, ammonium chloride and dry ice are some substances that ....................................
Fill in the blank
A ________ substance has only one kind of matter.
Fill in the blank
Clay is separated from water by the method called ______
Explain with diagram the process used to – separate the following substance from the given mixtures - Ammonium chloride from a mixture of – ammonium chloride & potassium chloride.
Explain with diagram the process used to – separate the following substance from the given mixtures - Carbon tetrachloride from a mixture of – carbon tetrachloride [heavier component] & water.
Name the apparatus that you will use to separate the components of mixtures containing two,
- miscible liquids,
- immiscible liquids
Lemonade is prepared by mixing lemon juice and sugar in water. You wish to add ice to cool it. Should you add ice to the lemonade before or after dissolving sugar? In which case would it be possible to dissolve more sugar?
Match the mixtures in column I with their method of separation in column II.
| COLUMN I | COLUMN II | ||
| (a) | Oil mixed in water |
(i) | Sieving |
| (b) | Iron powder mixed with flour | (ii) | Hand picking |
| (c) | Salt mixed with water | (iii) | Decantation |
| (d) | Lady’s finger mixed with french beans | (iv) | Magnet |
| (e) | Rice flour mixed with kidney beans | (v) | Evaporation |
