Advertisements
Advertisements
प्रश्न
Give a balanced equation for –
A displacement reaction involving a metal above hydrogen in the activity series with copper [II] sulphate solution
उत्तर
Displacement reaction
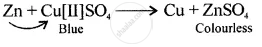
[more reactive displaces less reactive Cu]
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical equation for the following and identify the type of reaction.
\[\ce{Magnesium(s) + Hydrochloric acid(aq) -> Magnesium chloride(aq) + Hydrogen(g)}\]
State two characteristics of the chemical reaction which takes place when dilute sulphuric acid is poured over zinc granules.
What type of chemical reaction is represented by the following equation?
A + BC → AC + B
what type of reaction is the following:
Cl2 + 2KI → 2KCl + I2
What type of reaction is represented by the following equation?
Mg + CuSO4 → MgSO4 + Cu
What happens when a zinc strip is dipped into a copper sulphate solution?
Write the equation for the reaction that takes place.
What happens when a zinc strip is dipped into a copper sulphate solution?
Name the type of reaction involved.
Classify the following reaction into different type:
Zn + CuSO4 → ZnSO4 + Cu
Which of the following is a displacement reaction?
The colour of the solution observed after 30 minutes of placing zinc metal in copper sulphate solution is ______.
