Advertisements
Advertisements
प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
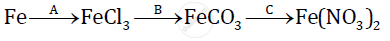
उत्तर
A:2Fe + 3Cl2 → 2FerCl3
B:2FeCl3 + 3Zn → 3ZnCl2 + 2Fe
Fe + H2CO3 → FeCO3 + H2 ↑
C: FeCO3 + 2HNO3 → Fe(NO3)2 + H2O+ CO2
APPEARS IN
संबंधित प्रश्न
Calculate the relative molecular mass of Sodium acetate
(use K = 39, Cl = 35.5, O = 16, C = 12, H = 1, Na = 23, N = 14, S= 32)
Empirical formula of a compound is CH2O. If its empirical formula is equal to its vapour density, calculate the molecular formula of the compound.
If a crop of wheat removes 20 Kg of nitrogen per hectare of soil, what mass of the fertilizer calcium nitrate,Ca(NO3)2 would be required to replace nitrogen in 10 hectare field? (N = 14, O = 16, Ca = 40)
When heated, potassium permanganate decomposes according to the following equation:
\[\ce{2KMnO4 -> \underset{solid residue}{K2MnO4 + MnO2} + O2}\]
Given that the molecular mass of potassium permanganate is 158 g, what volume of oxygen (measured at room temperature) would be obtained by the complete decomposition of 15.8 g of potassium permanganate? (Molar volume at room temperature is 24 litres). [K = 39, Mn = 55, O = 16]
A compound 'X' consists of 4.8% of C and 95.2% of Br by mass.
Name the type of chemical reaction by which X can be prepared from ethane.
Calculate the volume of oxygen required for the complete combustion of 8.8 g of propane (C3H5).
(Atomic mass: C = 14, O = 16, H = 1, Molar Volume = 22.4 dm3 at STP.)
Give one word or phrase for the following:
The ratio of the mass of a certain volume of gas to the mass of an equal volume of hydrogen under the same conditions of temperature and pressure.
Give two tests of the following:
Oxygen
Calculate the relative molecular mass of:
CuSO4. 5H2O
Find the weight of 0.5 mole of O2.
