Advertisements
Advertisements
प्रश्न
Give the disproportionation reaction of H3PO3
उत्तर
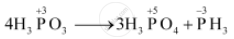
On heating, orthophosphorus acid (H3PO3) disproportionates to give orthophosphoric acid (H3PO4) and phosphine (PH3). The oxidation states of P in various species involved in the reaction are mentioned below.
APPEARS IN
संबंधित प्रश्न
What is the basicity of H3PO4?
Give reasons for the following : H3PO2 is a stronger reducing agent than H3PO3.
What are the oxidation states of phosphorus in H3PO3
What are the oxidation states of phosphorus in the following:
PCl3
Write balanced chemical equations involved in the following reactions:
Calcium phosphide is dissolved in water.
Which of the following acids forms three series of salts?
Which among the oxoacids of phosphorus contain two P – OH bonds, one P = O and one P – H bond
Ortho phosphoric acid on heating gives:-
Which one of the following is a dibasic acid?
Match List-I with List-II:
| List-I | List-II |
| name of oxo acid | Oxidation state of 'P' |
| (a) Hypophosphorous acid | (i) +5 |
| (b) Orthophosphoric acid | (ii) +4 |
| (c) Hypophosphoric acid | (iii) +3 |
| (d) Orthophosphorous acid | (iv) +2 |
| (v) +1 |
Choose the correct answer from the options given below:
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of H3PO4?
What is the basicity of \[\ce{H3PO4}\]?
What is the basicity of \[\ce{H3PO4}\]?
