Advertisements
Advertisements
प्रश्न
Give one equation to show the following property of sulphuric acid:
Acidic nature
उत्तर
Acidic nature of sulphuric acid: It acts as a strong dibasic acid.
\[\ce{H2SO4 -> 2Hi+ + SO^{2-}_{4}}\]
It reacts with metals, metal oxides, metal hydroxides, and carbonates to form metallic sulphates and hydrogen at ordinary temperature.
\[\ce{Mg + H2SO4 -> MgSO4 + H2\uparrow}\]
\[\ce{CuO + H2SO4 -> CuSO4 + H2O}\]
\[\ce{2NaOH + H2SO4 -> Na2SO4 + 2H2O}\]
\[\ce{ZnCO3 + H2SO4 -> ZnSO4 + H2O + CO2\uparrow }\]
APPEARS IN
संबंधित प्रश्न
Give reason for the following:
H2SO4 cannot be prepared by heating sodium sulphate with conc. HCl or HNO3
Give examples of the use of sulphuric acid as
Non-volatile acid
Choose the current answer from the options given below :
When conc. H2SO4 comes in contact with sugar, it becomes black due to
Name the catalyst used in the process.
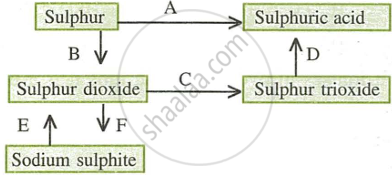
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
State your observation for the following case :
Paper soaked in potassium permanganate solution is introduced into a gas jar of sulphur dioxide.
Write the balanced chemical equation for the following conversion:
Ammonium sulphate from ammonia and dilute sulphuric acid.
Give a chemical test to distinguish between dilute sulphuric acid and dilute hydrochloric acid, (using lead nitrate solution).
A, B and C are the chemical properties of sulphuric acid:
A. Oxidizing agent
B. Dehydrating agent
C. Non-volatile acid
Match the following equations 1 to 3 to the above chemical properties of sulphuric acid.
1. \[\ce{KNO3 + H2SO4 -> KHSO4 + HNO3}\]
2. \[\ce{C12H22O11 -> 12C + 11H2O}\]
3. \[\ce{S + 2H2SO4 -> 2H2O + 3SO2}\]
Differentiate between the following pair based on the criteria given in the bracket:
Cane sugar and hydrated copper sulphate [using concentrated H2SO4]
