Advertisements
Advertisements
प्रश्न
Give one example of isobars.
उत्तर
`""_11^23Na` and `""_12^23Mg` are isobar having same mass number 23 and different atomic numbers.
संबंधित प्रश्न
What do you mean by Atomic number
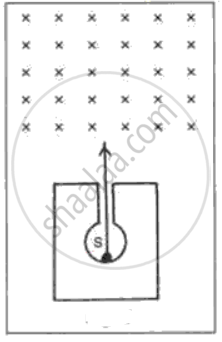
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
Draw a diagram of a simple atom showing the nucleus and electrons. In this atom:
(a) What type of charge will there be on the nucleus?
(b) What is the value of this charge?
A radioactive substance is oxidized. What changes would you expect to take place in the nature of radioactivity? Explain your answer.
State three safety precautions that you would take while handling the radioactive substances.
Why should a radioactive substance not be touched by hands?
State two properties that a substance should possess when used as a thermionic emitter.
Are all the radiations viz. α, β and γ emitted in a single radioactive decay?
Which of the following statements is/are correct?
- α particles are photons
- Penetrating power of γ radiation is very low
- Ionization power is maximum for α rays
- Penetrating power of γ radiation is very high
In the following atoms, which one is a radioisotope? Give one use of this isotope.
O16, C14, N14, He4
